Fujifilm Sonosite Unveils Next-Generation Point-of-Care Ultrasound Device | Diagnostic Imaging
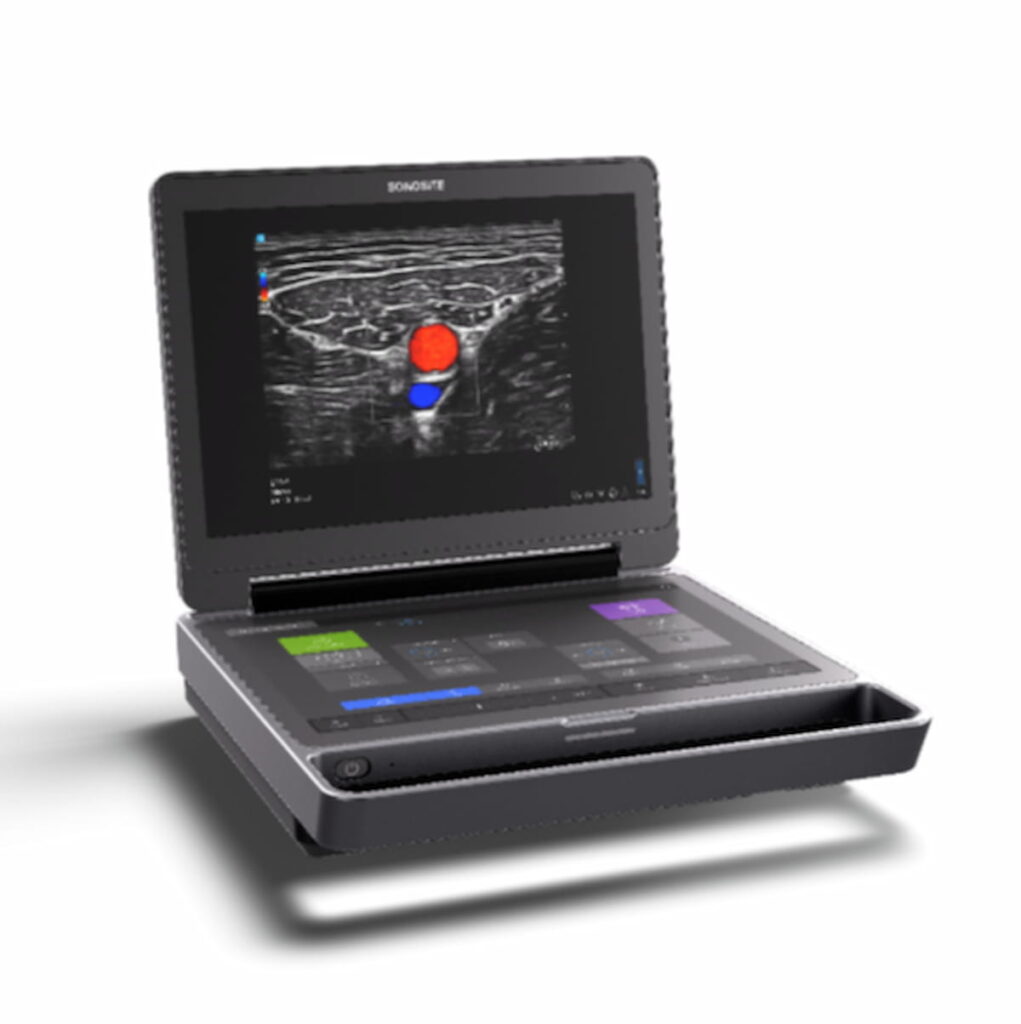
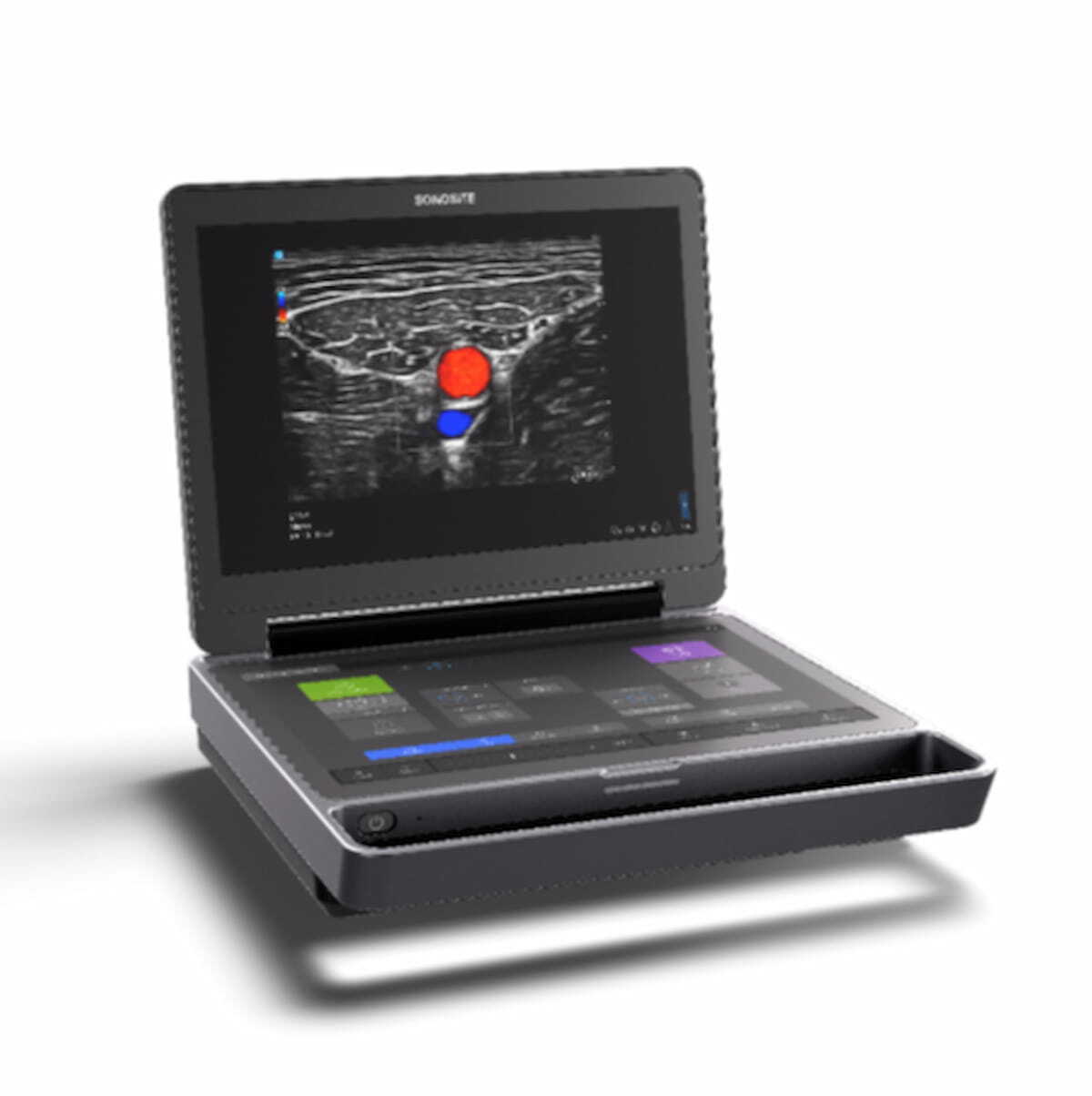
The Sonosite MT ultrasound device reportedly combines portability and enhanced high-resolution images with multiple features to promote workflow efficiency.
Read the full article at: www.diagnosticimaging.com
RapidAI Garners Four New FDA Clearances For CT-Based AI Modules | Diagnostic Imaging
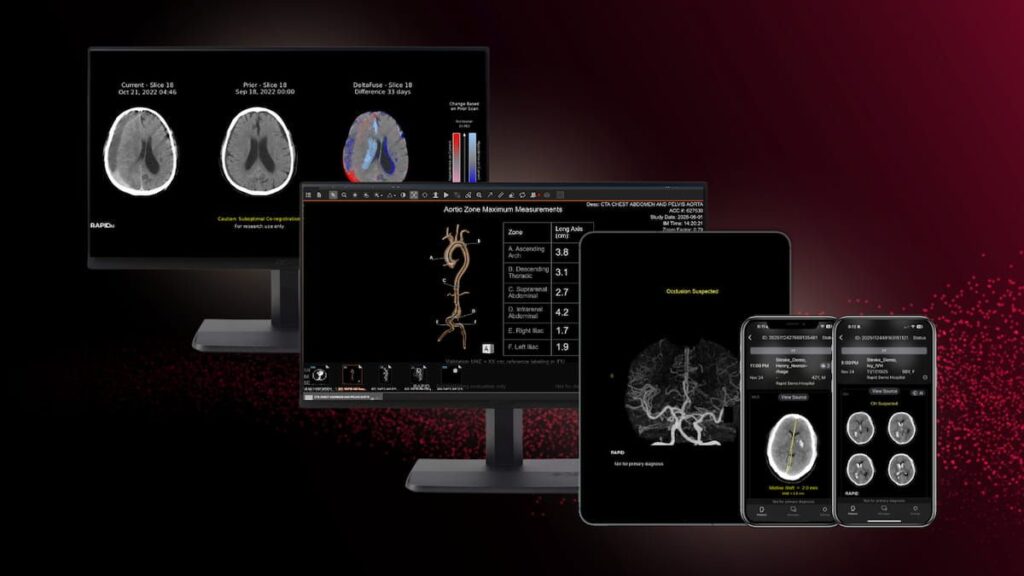
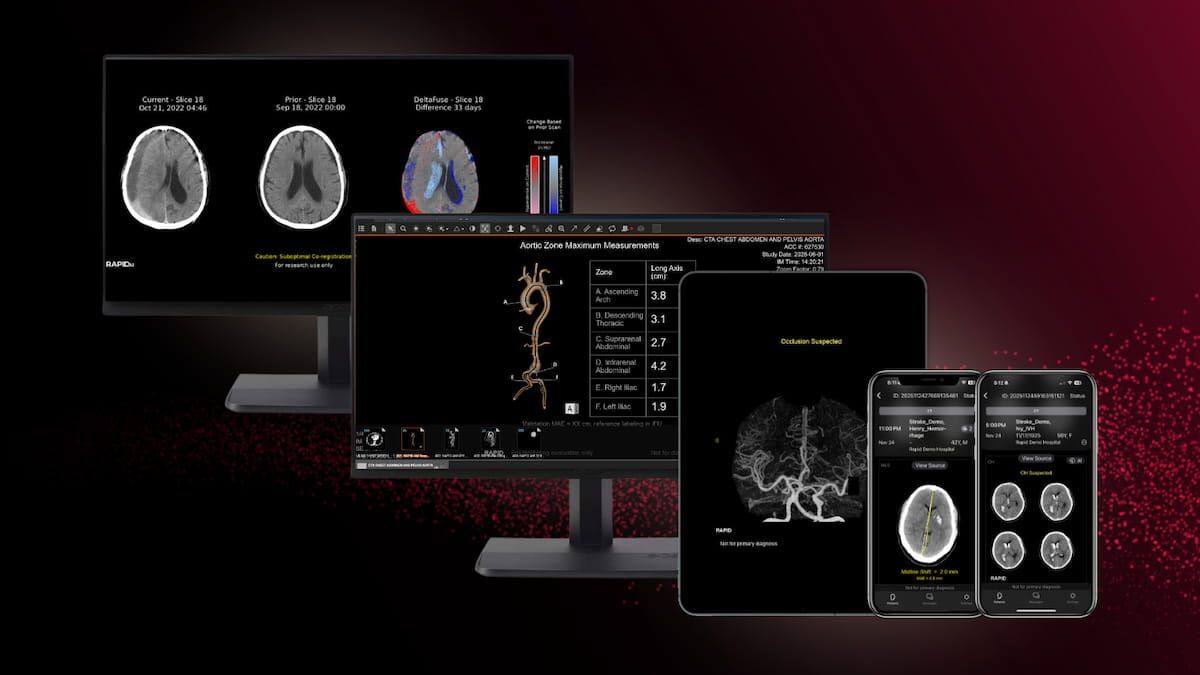
The new FDA 510(k) clearances for RapidAI include Rapid LMVO, which facilitates assessment of ischemic stroke, and Rapid MLS, which aids in the quantification of midline shifts with potential brain injuries.
Read the full article at: www.diagnosticimaging.com
FDA Data Reveals Medical Device Recalls Software Woes


Discover how FDA medical device recall data exposes software design as a key factor in device recalls, impacting healthcare quality and patient assurance.
Read the full article at: spectrum.ieee.org
Ultrasound Renal Denervation System for Hypertension Garners CMS National Coverage Determination | Diagnostic Imaging
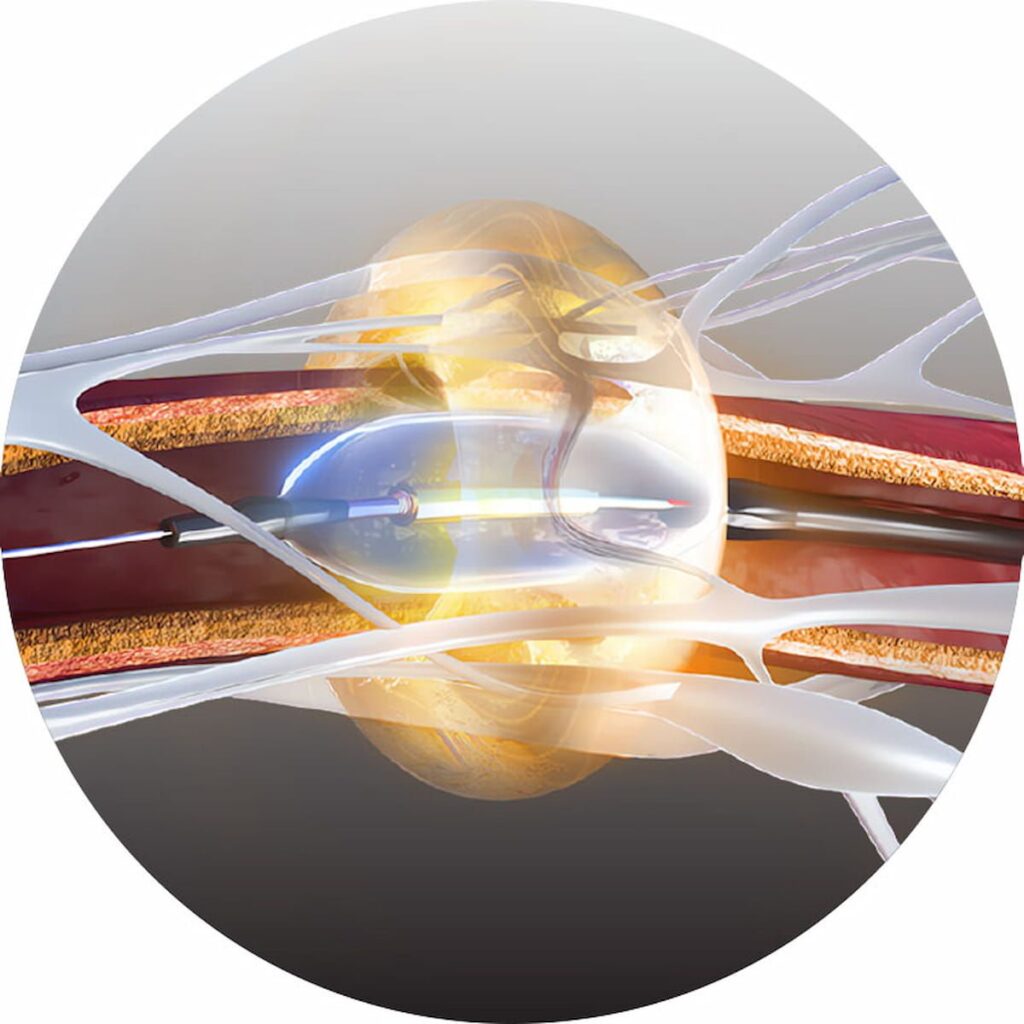
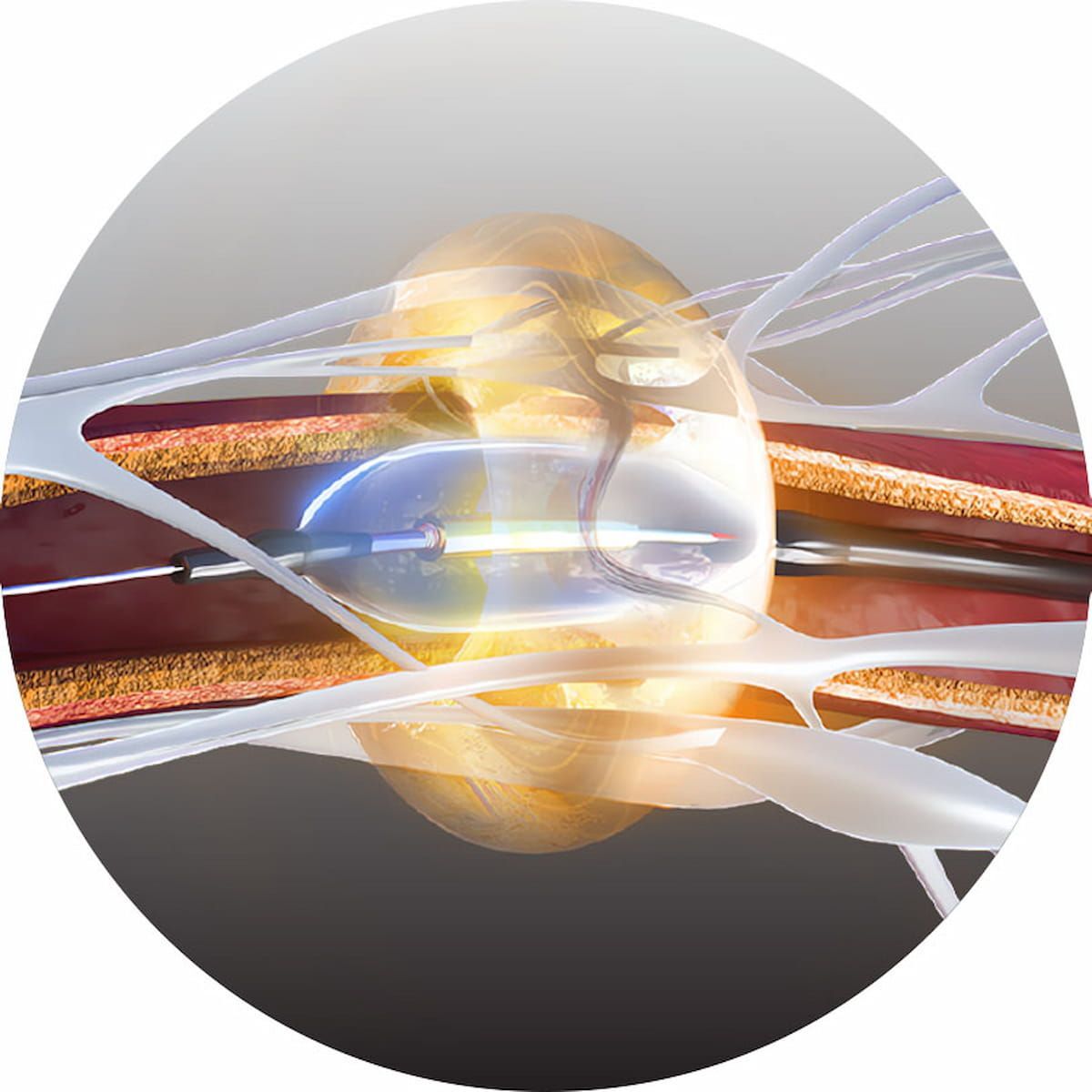
The Paradise Ultrasound Renal Denervation System has demonstrated efficacy and a favorable safety profile in randomized controlled trials for the treatment of mild, moderate and resistant hypertension.
Read the full article at: www.diagnosticimaging.com
Emerging AI Software for CT Plaque Analysis ‘Revolutionizes’ Carotid Artery Assessment | Diagnostic Imaging
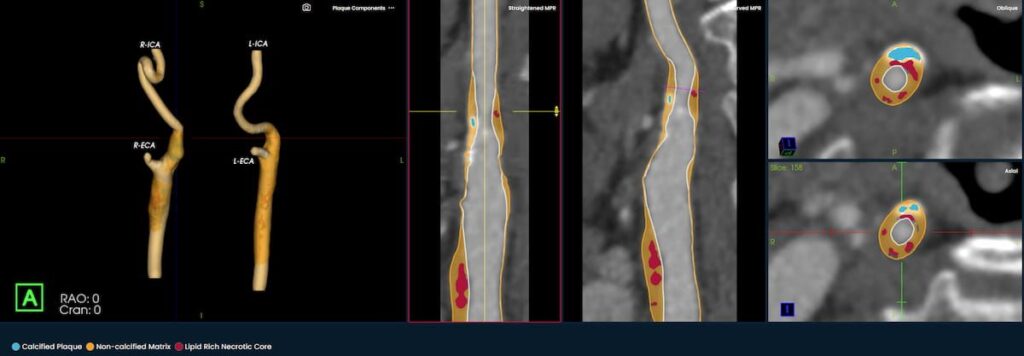
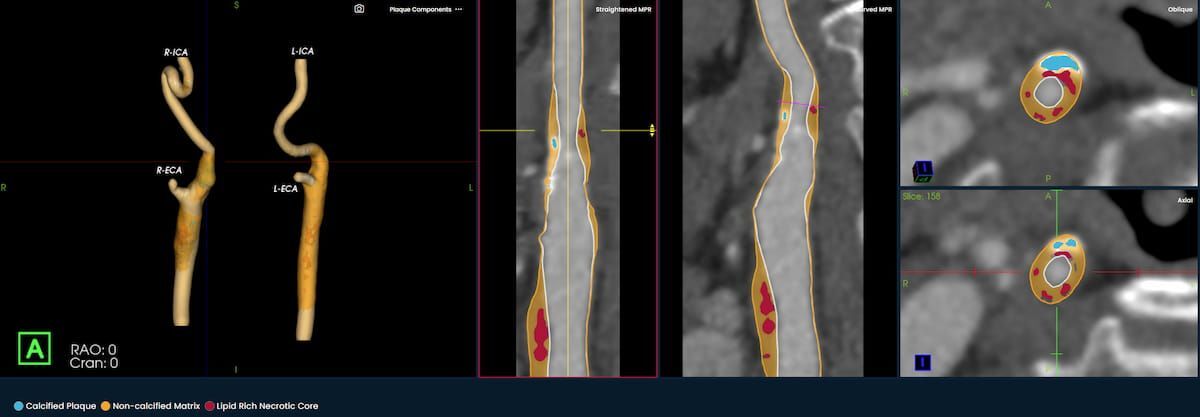
The newly launched PlaqueIQ™ image analysis modality is reportedly the first CT-based software indicated for the quantification and classification of plaque morphology in the carotid arteries.
Read the full article at: www.diagnosticimaging.com
BD Receives FDA 510(k) Clearance and CE-IVDR Certification for High-Throughput Enteric Bacterial Panels on BD COR™ System


/PRNewswire/ — BD (Becton, Dickinson and Company) (NYSE: BDX) a leading global medical technology company, today announced it has received U.S. Food and Drug…
Read the full article at: www.prnewswire.com
When Did the FDA Begin Regulating UVC Robots?
Infection Control Today serves infection control, facility, and C-suite leaders with strategies on HAIs, patient care, safety, and quality outcomes…
Read the full article at: www.infectioncontroltoday.com
QuidelOrtho Receives FDA 510(k) Clearance for VITROS™ Immunodiagnostic Products hs Troponin I Assay


/PRNewswire/ — The U.S. Food and Drug Administration (“FDA”) has granted QuidelOrtho Corporation (Nasdaq: QDEL) (“QuidelOrtho”), a global leader of in vitro…
Read the full article at: www.prnewswire.com
Tosoh Bioscience Receives FDA 510(k) Clearance for Fast, Accurate GR01 HbA1c Testing Analyzer


/PRNewswire/ — Tosoh Bioscience, Inc., a market leader in clinical diagnostics has received U.S. FDA 510(k) clearance for its next-generation Tosoh Automated…
Read the full article at: www.prnewswire.com
Clinical Trials and Regulatory Reality: Essential Planning for FDA Success
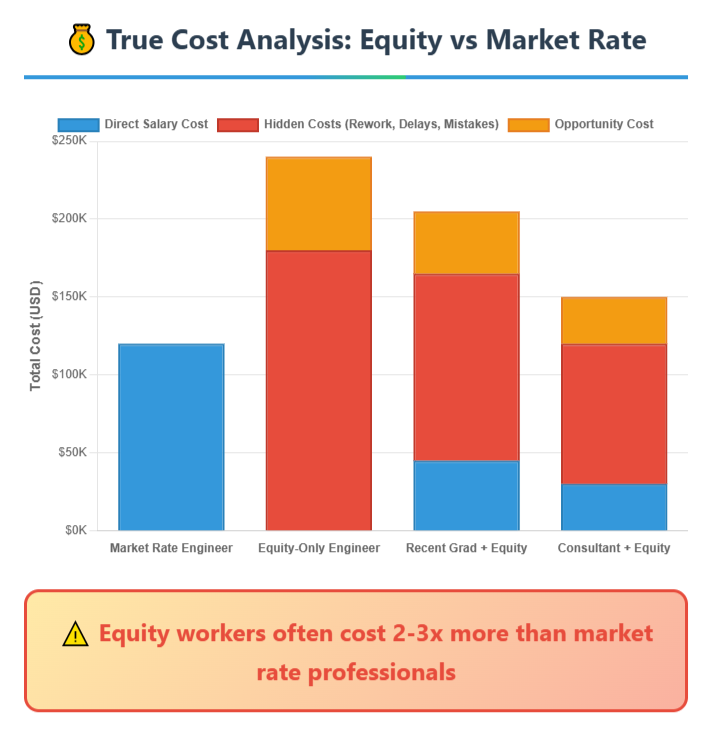
“The cheapest team member might be the most expensive mistake you make.


