Hologic Receives FDA Clearance and CE Mark for Automated Molecular Tests to Detect Common Causes of Infectious Gastroenteritis
MARLBOROUGH, Mass.–(BUSINESS WIRE)—- $HOLX #Bacterial–Hologic, Inc. (Nasdaq: HOLX) announced today that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the Panther Fusion® Gastrointestinal (GI) Bacterial and Expanded Bacterial Assays. The company also announced that the assays obtained CE marking in the European Union in accordance with In Vitro Diagnostic Regulation (IVDR). These highly sensitive molecular tests are designed to rapidly detect the most common bacterial pathogens res
Read the full article at: www.businesswire.com
Synchrony Medical Launches LibAirty™ Airway Clearance System in the U.S., Offering a New Standard for At-Home Respiratory Care


/PRNewswire/ — Synchrony Medical, a medical technology company dedicated to transforming respiratory care, announced today the launch of its LibAirty™ Airway…
Read the full article at: www.prnewswire.com
Biolinq gets FDA de novo nod for intradermal glucose sensor


The device, called Biolinq Shine, is indicated for people with Type 2 diabetes who don’t depend on insulin.
Read the full article at: www.medtechdive.com
Smartlens, Inc. Secures $5.2 Million Bridge Round to Advance FDA Clearance Process and Prepare for Commercial Launch of miLens


/PRNewswire/ — Smartlens, Inc., a clinical-stage ophthalmic technology company advancing next-generation glaucoma management devices, today announced the…
Read the full article at: www.prnewswire.com
Auxano® Medical Receives FDA 510(k) Clearance for the ARKEO™ Wedge Fixation System – Ortho Spine News


An important milestone in the company’s mission to deliver precision-engineered orthopedic solutions BRECKSVILLE, OH, UNITED STATES, October 16, 2025 /EINPresswire.com/ — Auxano® Medical is proud to announce its wedge fixation system, ARKEO™, has received U.S. Food and Drug Administration (FDA) 510(k) clearance, marking an important milestone in the company’s mission to deliver precision-engineered orthopedic solutions that empower surgeons and improve patient outcomes. The ARKEO™ Wedge Fixation System integrates an anatomical design with the pillared microstructure of the Auxano® in-growth technology to provide a secure fixation interface at the site of correction. Designed for use in Evans and Cotton osteotomies, the ARKEO™ surgical set includes intuitive instrumentation that enhances reproducibility by giving surgeons complete control over implant positioning throughout the procedure. “Achieving 510(k) clearance for ARKEO™ represents a significant advancement in our orthopedic portfolio,” said Joe Harr, Sales & Distribution Manager at Auxano® Medical. “This system combines modern engineering with practical, surgeon-focused innovation to deliver reproducible results and improve the surgical experience.” The ARKEO™ system is designed to optimize fixation and graft integration through several key features: • Anatomical Fit: Subflush placement reduces irritation and maintains alignment. • Open Architecture: Large central graft cavity and thru-growth fenestrations maximize graft capacity and promote biologic fixation. • Controlled Stability: The unique pillared surface morphology has demonstrated viability in scientific research studies and clinical efficacy in midfoot fusion applications. • Intuitive Instrumentation: Simplifies each step for efficient, consistent outcomes. Supplied non-sterile in a steam-sterilizable tray, the ARKEO™ system includes a Hintermann distractor, k-wires, osteotomes, mallet, slap hammer, implant trials, inserter, and an impactor. “The ARKEO™ Wedge Fixation System exemplifies Auxano® Medical’s commitment to designing devices that are both clinically advanced and intuitively engineered,” shared Dr. George Picha, Ph.D, F.A.C.S. “With this clearance, we are proud to continue expanding our solutions for orthopedic surgeons across the U.S.” For more information about the ARKEO™ Wedge Fixation System, visit www.AuxanoMedical.net or contact CS@AuxanoMedical.net.About Auxano® Medical Auxano® Medical develops and manufactures advanced orthopedic solutions that support improved patient outcomes and procedural efficiency. With a commitment to quality, innovation, and clinical collaboration, Auxano® delivers technologies that meet the highest standards of performance and reliability. The company is headquartered near Cleveland, OH. For more information, visit www.auxanomedical.net Halle SchosslerAuxano Medicalemail us here Legal Disclaimer: EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Read the full article at: orthospinenews.com
FDA Green Lights Ruthlessly Practical ‘Smart’ Device

Spine surgeons know the drill: precision is everything when it comes to pedicle screw placement.
Read the full article at: ryortho.com
FDA halts new device submissions during government shutdown


The FDA will not accept new medical device submissions that require payment of user fees during the Trump administration’s government shutdown.
Read the full article at: www.medicaldesignandoutsourcing.com
Next-Generation AI-Driven SyncAR® Spine Receives FDA Clearance for Spine Surgery –
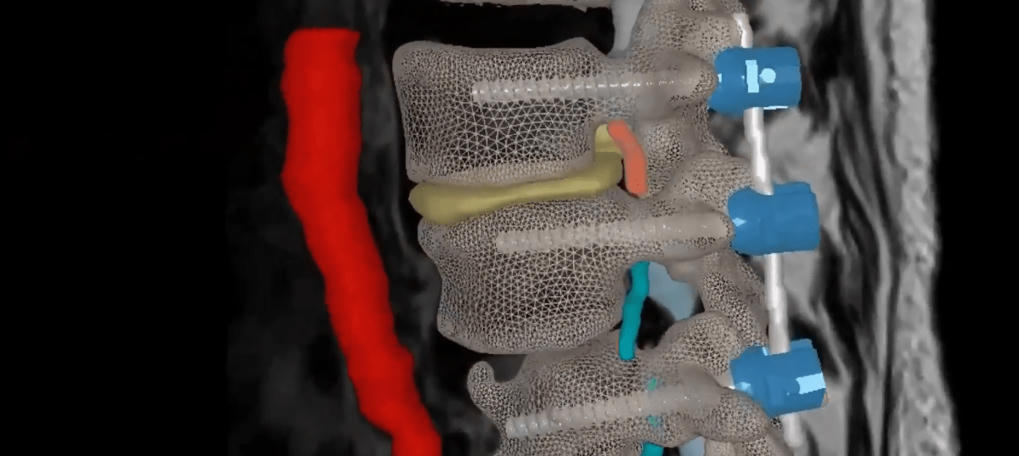

Surgical Theater, the leader in surgical XR visualization, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for SyncAR® Spine, the next-generation release of its spine platform.
Read the full article at: orthofeed.com
Versatile Full-Body Cone-Beam CT Scanner Gets FDA Clearance
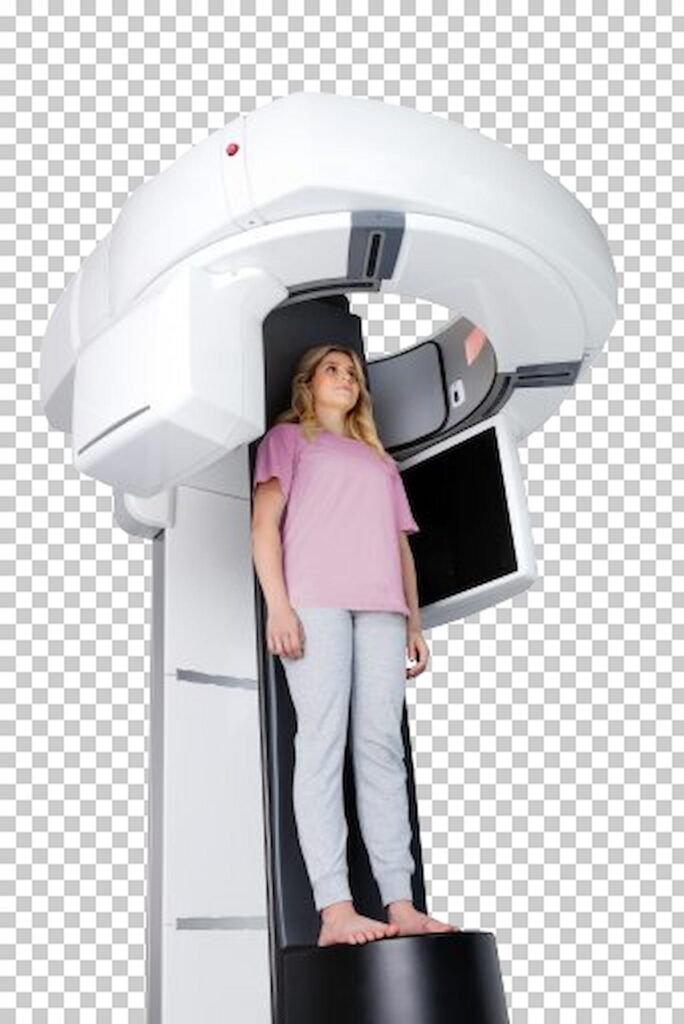

The Planmed XFI full-body weightbearing cone-beam CT scanner reportedly enables upright and supine patient positioning.
Read the full article at: www.diagnosticimaging.com
Prytime Medical Devices Receives World’s First Extended Duration FDA 510(k) Clearance for pREBOA-PRO™ Catheter


/PRNewswire/ — Building once again on their first mover position as a leader in endovascular hemorrhage control and resuscitation, Prytime Medical Devices,…
Read the full article at: www.prnewswire.com


