Francis Medical gets FDA green light for water vapor ablation to treat prostate cancer


Francis Medical garnered a 510(k) clearance for its water vapor ablation therapy for cancers of the prostate, kidneys and bladder.
Read the full article at: www.fiercebiotech.com
Joint Preservation Innovations Announces FDA 510(k) Clearance for the Articulator™ Arthroscopic Bur — the First and Only FDA-Cleared Articulating Rotary Cutting Instrument


Joint Preservation Innovations Inc. announced that FDA has granted 510(k) clearance for the Articulator™ Arthroscopic Bur.
Read the full article at: ryortho.com
FDA making use of real-world evidence in device applications easier (BSX:NYSE)


Learn how the FDA is easing real-world evidence rules for medical devices, reducing data burdens for sponsors.
Read the full article at: seekingalpha.com
Atraverse Medical Secures FDA 510(k) Clearance for HOTWIRE™ RF Generator & Fully Integrated Transseptal Access System
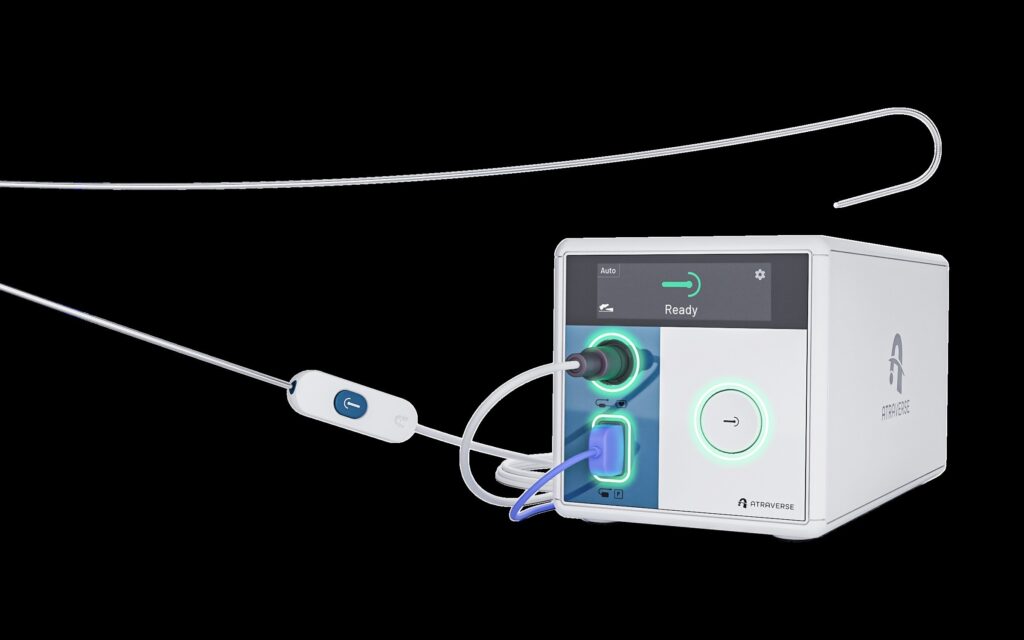
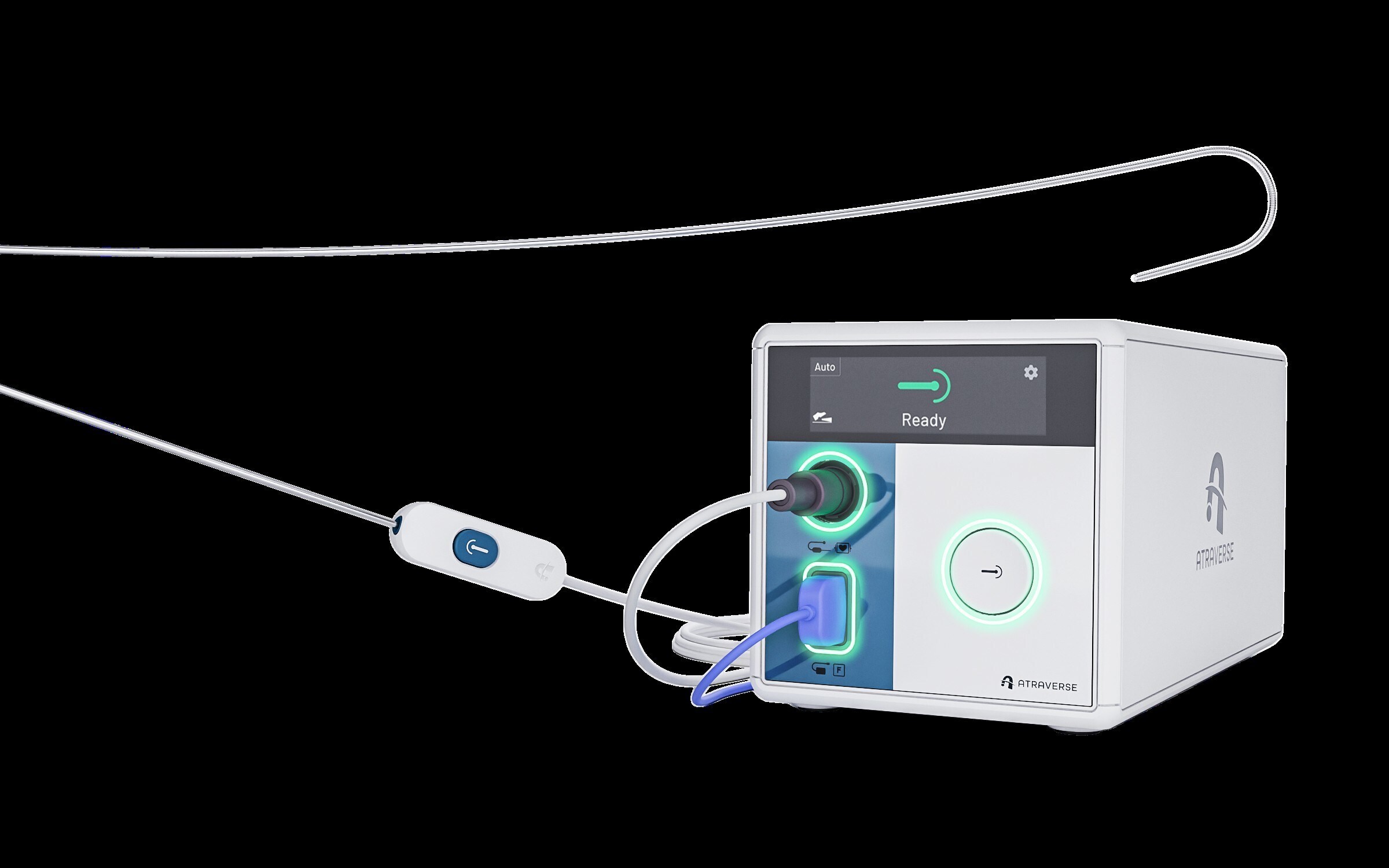
/PRNewswire/ — Atraverse Medical, a medical device company pioneering next-generation left-heart access technology, has received 510(k) clearance from the U.S….
Read the full article at: www.prnewswire.com
Galway medtech SymPhysis Medical seeks FDA approval


Galway medtech firm SymPhysis Medical is looking to complete a crowd-funding round by the end of the year, as part of up to €5m of investment sought to help bring its key medical device to market.
Read the full article at: www.independent.ie
Cutera® Announces New FDA Clearances for truFlex® to Support Rehabilitation, Recovery and Muscle Wellness


/PRNewswire/ — CUTERA, INC., a leading provider in aesthetic, dermatological solutions, today announced that the U.S. Food and Drug Administration (FDA) has…
Read the full article at: www.prnewswire.com
FDA Clears Ceribell’s AI-Powered EEG as First Device for Continuous Delirium Monitoring


Ceribell, Inc.has secured FDA 510(k) clearance for the world’s first continuous, AI-powered delirium monitoring solution,…
Read the full article at: hitconsultant.net
Bendit Announces FDA Clearance for the Bendit17™ Steerable Microcatheter


/PRNewswire/ — Bendit Technologies is proud to announce that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the Bendit17™…
Read the full article at: www.prnewswire.com
MMI earns FDA clearance for robotic soft tissue dissection tools
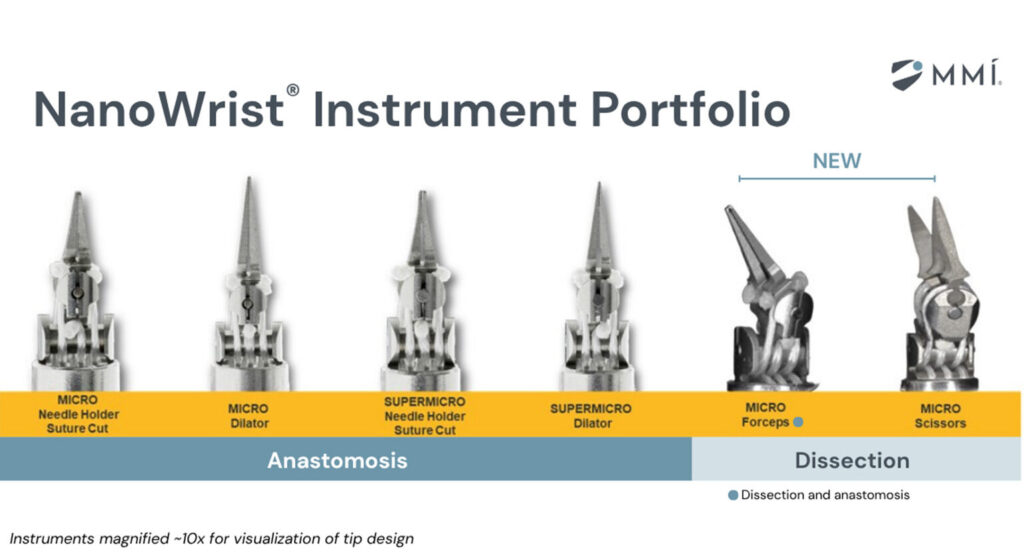
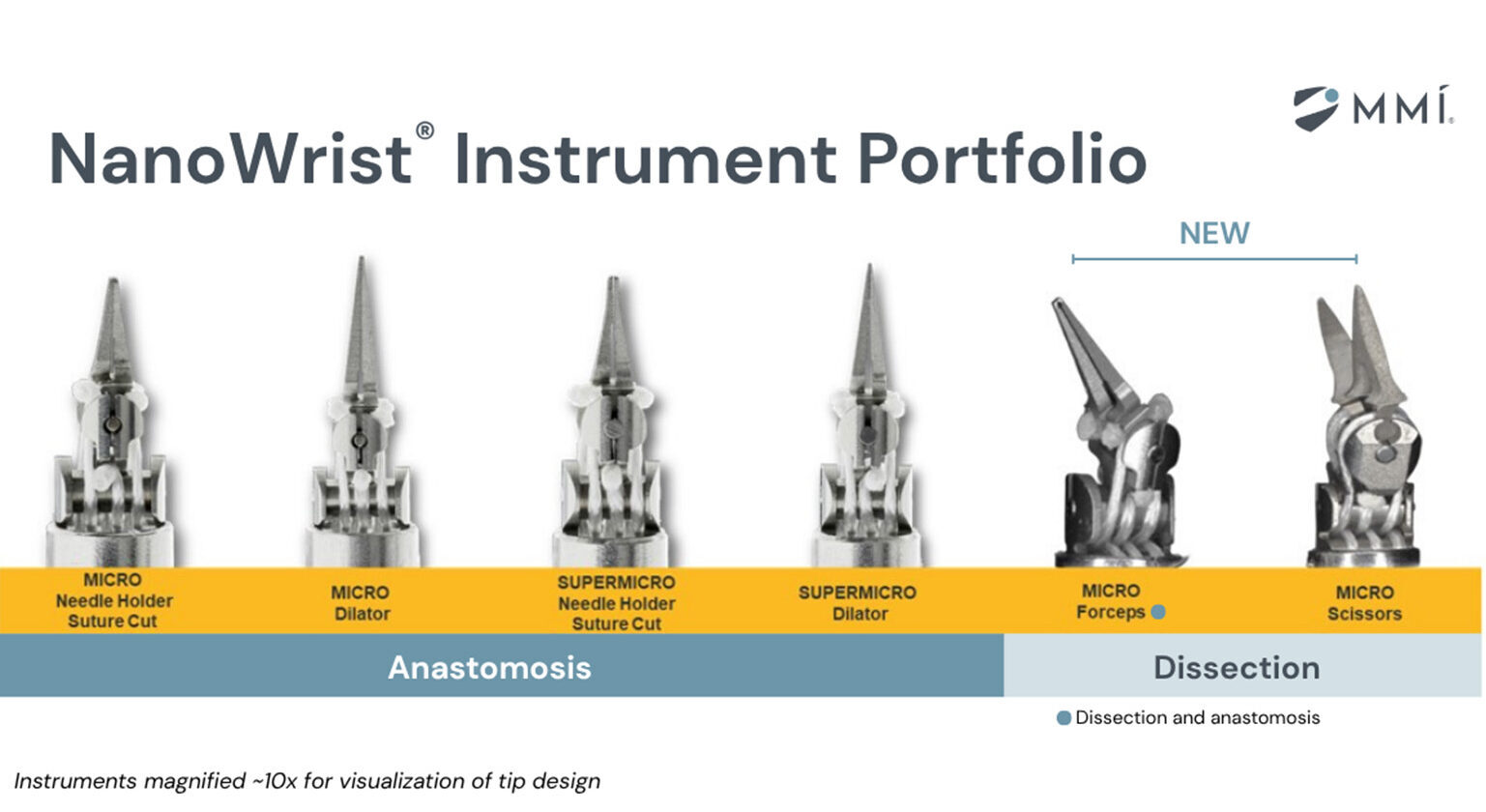
MMI’s dissection instruments are some of the smallest fully wristed robotic instruments designed for dissection in open microsurgery.
Read the full article at: www.therobotreport.com
Coronary thrombectomy system gains US FDA clearance
Vesalio has announced US Food and Drug Administration (FDA) 510(k) clearance and the upcoming US commercial launch of enVast, a clot retriever specifically cleared for mechanical thrombectomy in the cardiac circulation.
Read the full article at: cardiovascularnews.com


