SS Innovations submits robotic system for FDA clearance


After introducing its robot in multiple countries outside the U.S., including India, the company turns its focus to the world’s largest robotic surgery market.
Read the full article at: www.medtechdive.com
Medivis receives “world’s first” US FDA clearance for AR navigation in cranial neurosurgery
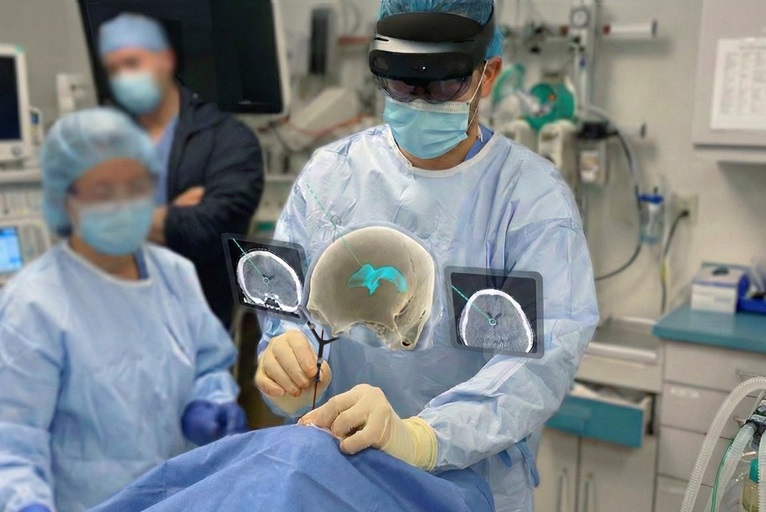
Medivis has announced that it has received US Food and Drug Administration (FDA) 510(k) clearance for its cranial navigation platform, making it the “world’s first” augmented reality (AR) system cleared for intraoperative guidance in cranial neurosurgery, according to the company.
Read the full article at: neuronewsinternational.com
HeartBeam swells on turning FDA rejection into ECG clearance


The FDA’s change of heart has HeartBeam planning a commercial launch in the new year for its credit card-sized ECG system.
Read the full article at: www.fiercebiotech.com
CMR Surgical receives FDA clearance for Versius Plus surgical robot
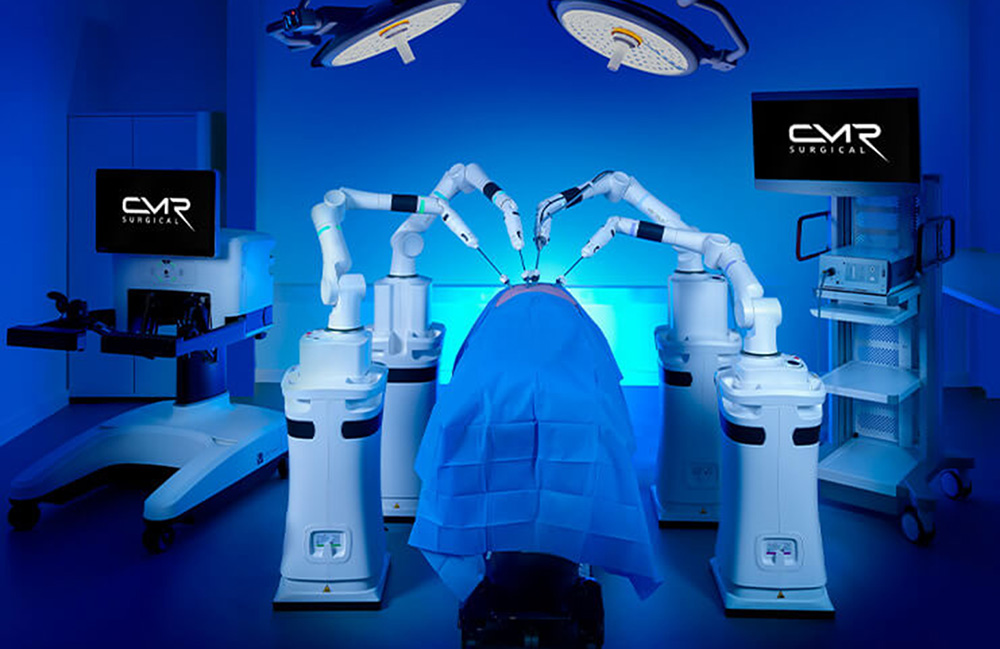
The FDA previously granted marketing authorization for CMR’s first-generation Versius Surgical System in October 2024.
Read the full article at: www.therobotreport.com
FDA OKs First At-Home Brain Stim Device for Depression


The FL-100 tDCS system is available by prescription for adults with moderate to severe MDD in the current episode, either as monotherapy or as an adjunctive treatment.
Read the full article at: www.medscape.com
Withings Redefines At-Home Family Healthcare in the US with FDA Clearance of BeamO
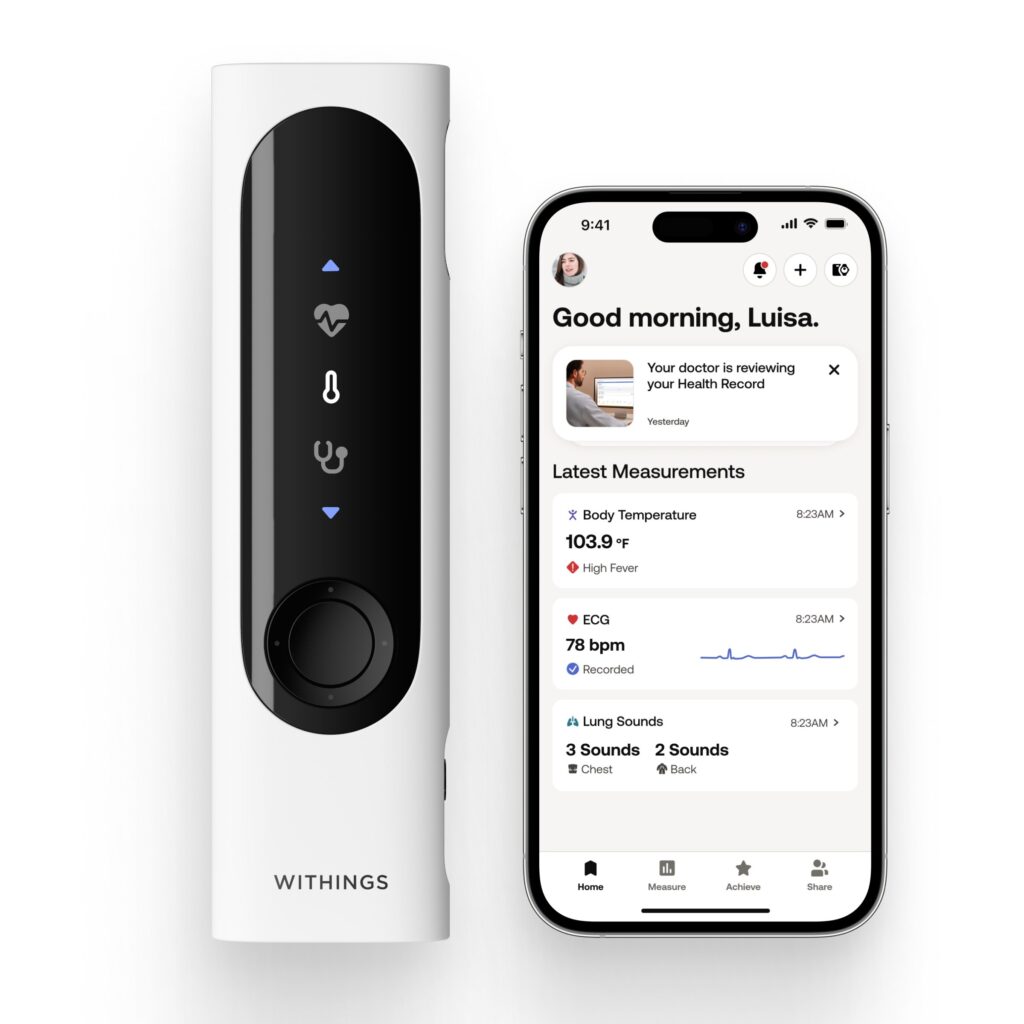
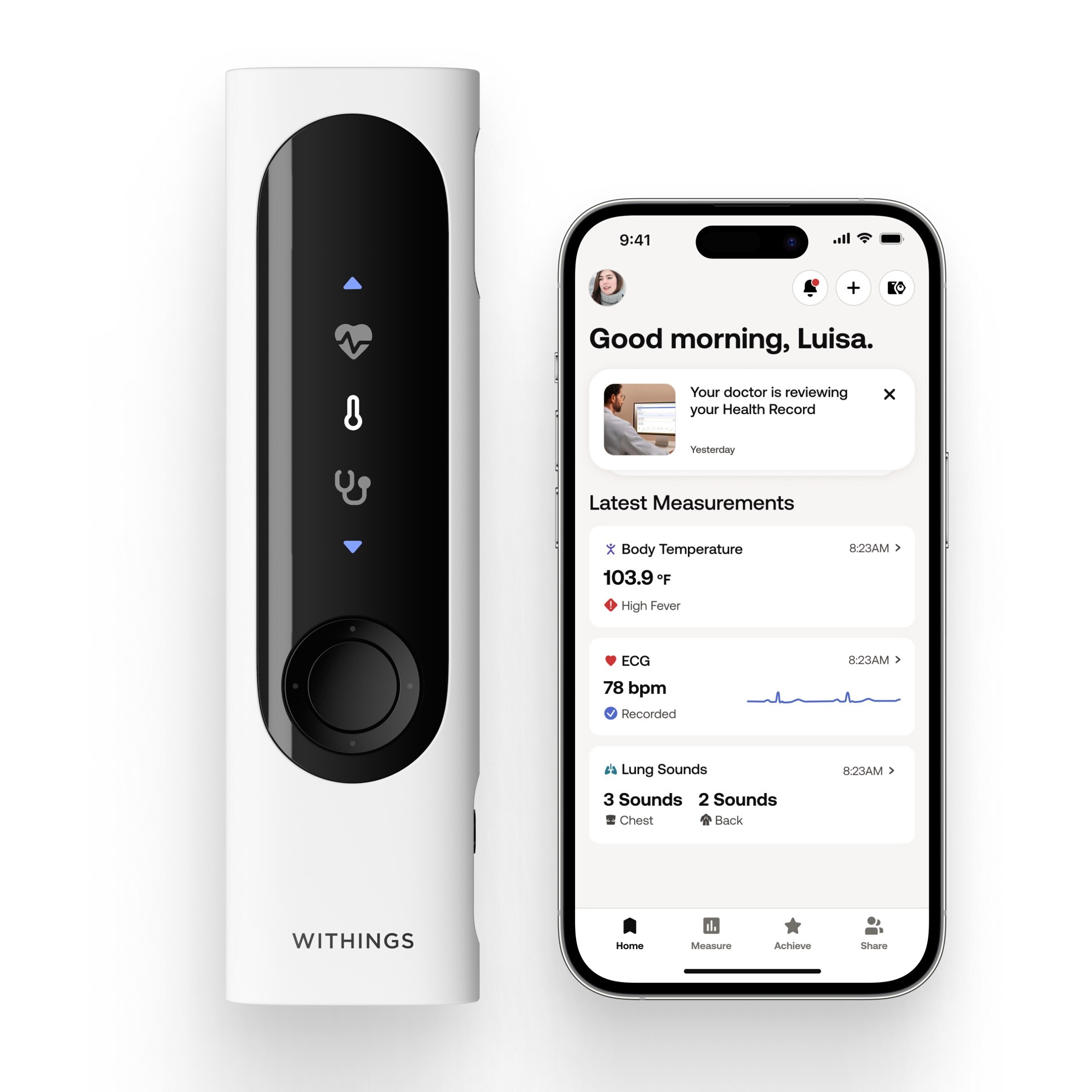
/PRNewswire/ — Withings, the world leader in connected health, today announced that the FDA has cleared BeamO1, the revolutionary health check-up tool. This…
Read the full article at: www.prnewswire.com
Mindray Debuts Resona A20 Premium Ultrasound Device at RSNA | Diagnostic Imaging
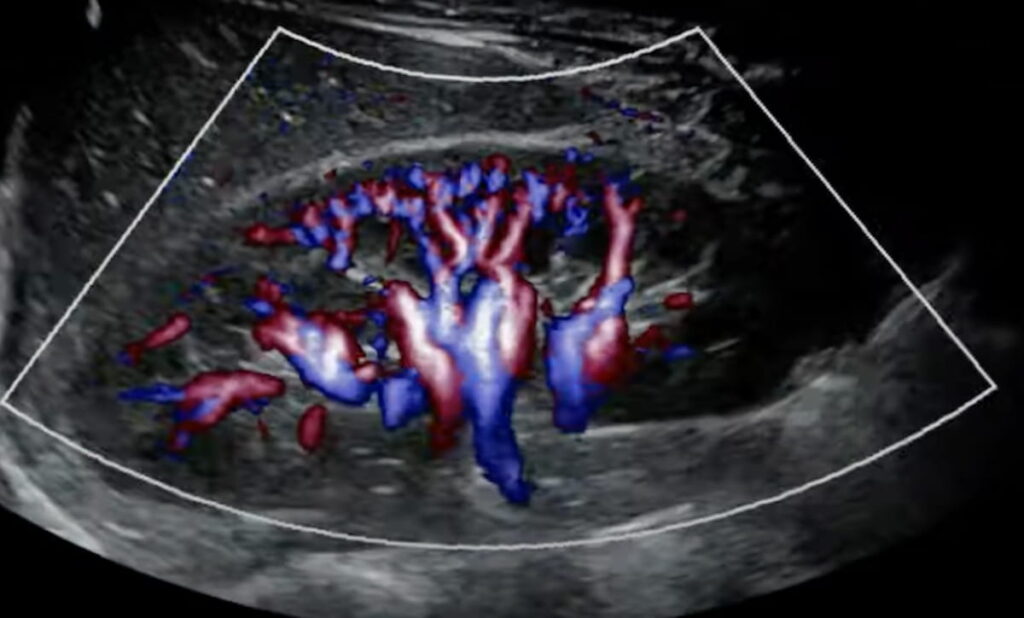
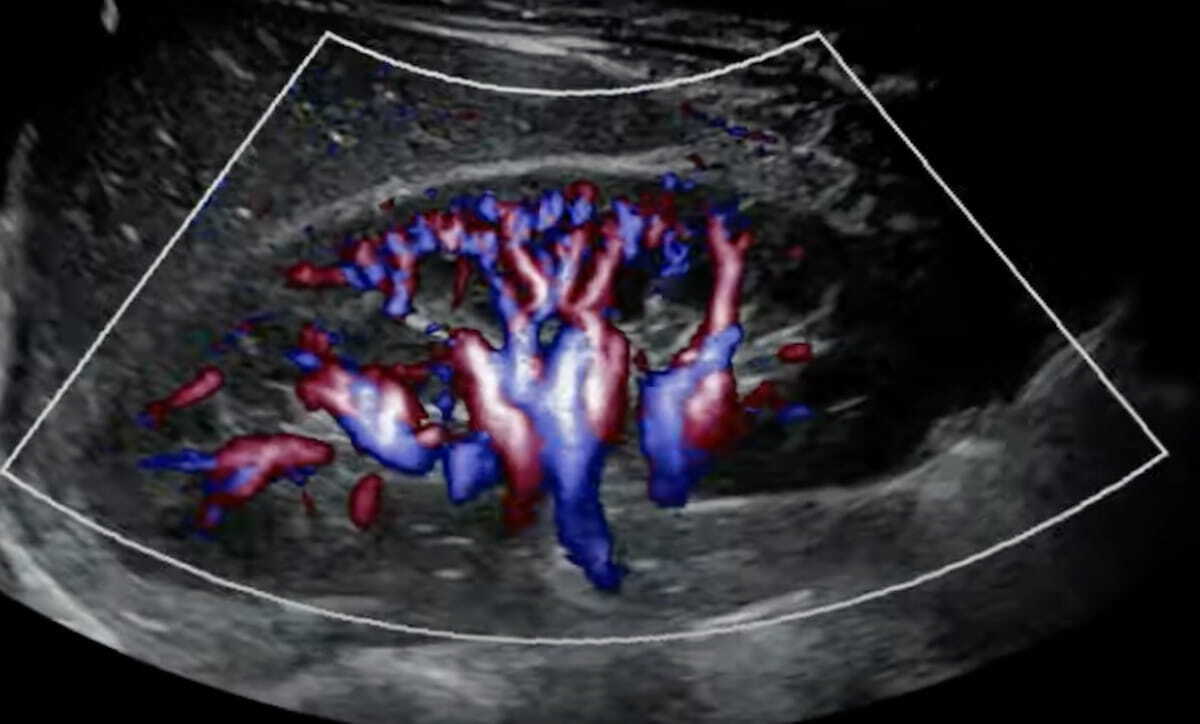
The Resona A20 device reportedly provides advances in imaging and AI automation along with an intuitive ergonomic design for ease of use.
Read the full article at: www.diagnosticimaging.com
Paradromics Gets FDA Approval to Trial Its Brain Implant in People


The Austin-based startup will test its high-bandwidth device to help restore speech in people with extremely limited movement.
Read the full article at: www.wired.com
Augmedics wins FDA clearance for next-gen AR headset


Augmedics announced today that it received FDA 510(k) clearance for X2, its next-generation augmented reality (AR) headset.
Read the full article at: www.massdevice.com
Stereotaxis nets FDA clearance for redesigned surgical robot


The company also posted weaker third-quarter results, but CEO David Fischel said orders for the new GenesisX system, when fully launched, would outpace the rate for an older model.
Read the full article at: www.medtechdive.com


