Rapid Nexus Becomes the First Company to Receive FDA Clearance for its Advanced Wound-Healing Device Hemastyl


/PRNewswire/ — Rapid Nexus Nanotech Wound Solutions, Inc., a California-based med-tech company focused on advanced wound care, today announced it has received…
Read the full article at: www.prnewswire.com
Zimmer Biomet Receives FDA Clearance For Enhanced Knee Replacement Technology –
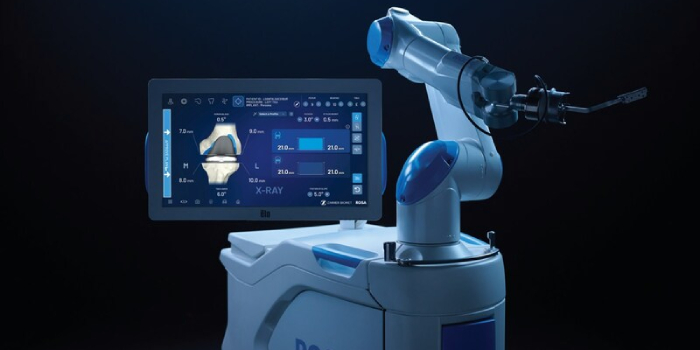
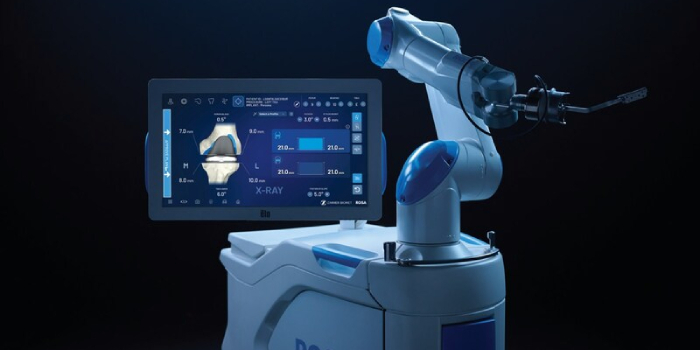
Warsaw-based Zimmer Biomet, a global medical technology leader, today announced U.S. Food and Drug Administration, clearance of ROSA Knee with OptimiZe, an enhanced version of its ROSA Knee System that offers a more customized experience for surgeons to help deliver accurate and reproducible…
Read the full article at: www.inkfreenews.com
Vesalio receives two US FDA 510(k) clearances for aspiration devices


Vesalio recently announced two new US Food and Drug Administration (FDA) 510(k) clearances for its aspiration devices.
Read the full article at: neuronewsinternational.com
Meduloc Steps Into the Spotlight With FDA Clearance
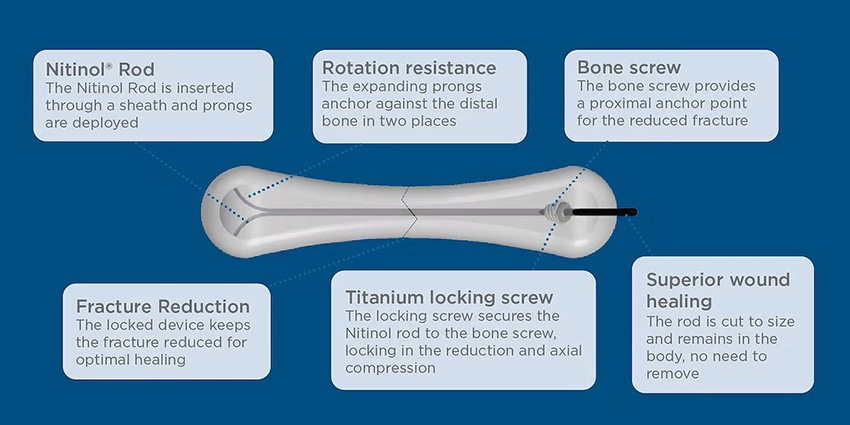
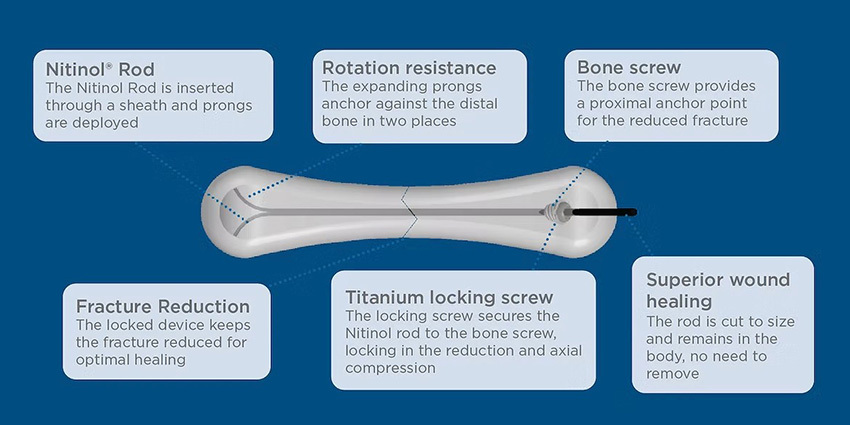
FDA just cleared a fresh option for small bone fixation which is minimally invasive, fast, stable, and more patient-friendly.
Read the full article at: ryortho.com
IR Update: FDA Clears Emerging Prostate Tissue Ablation System for Intermediate Risk PCa | Diagnostic Imaging

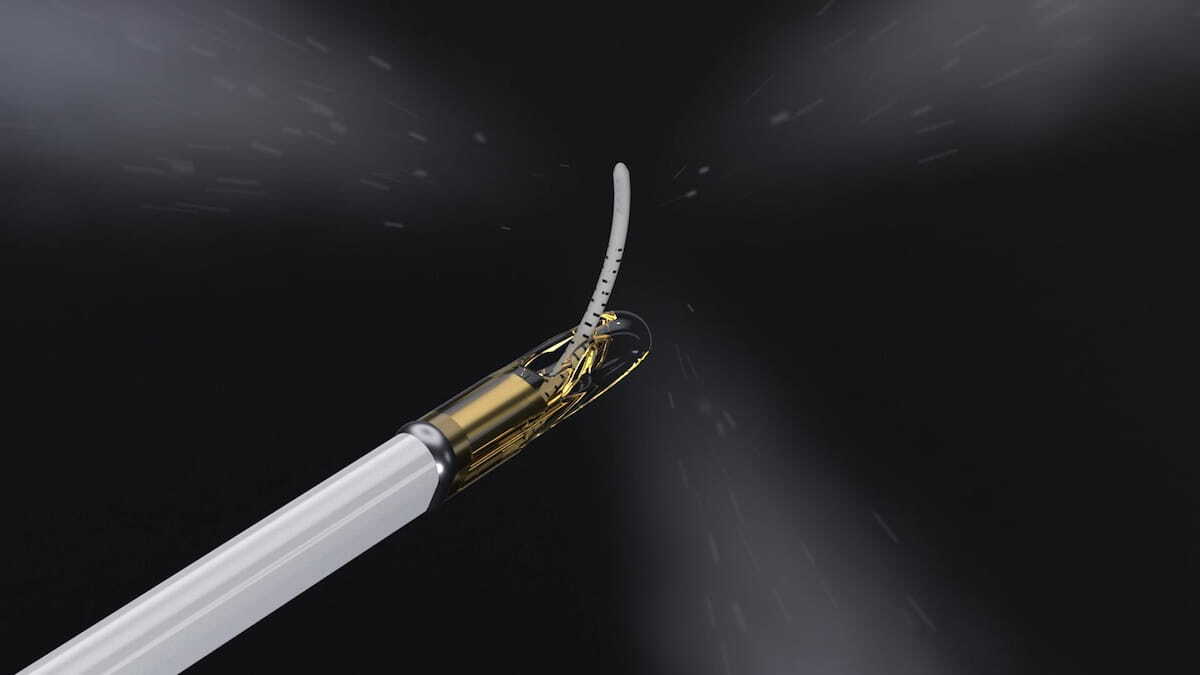
Preliminary research has shown that the Vanquish Water Vapor Ablation System eliminated MRI-visible intermediate-risk PCa in over 90 percent of patients.
Read the full article at: www.diagnosticimaging.com
Integra gains FDA clearance to use CUSA Clarity Aspirator for cardiac surgeries (IART:NASDAQ)
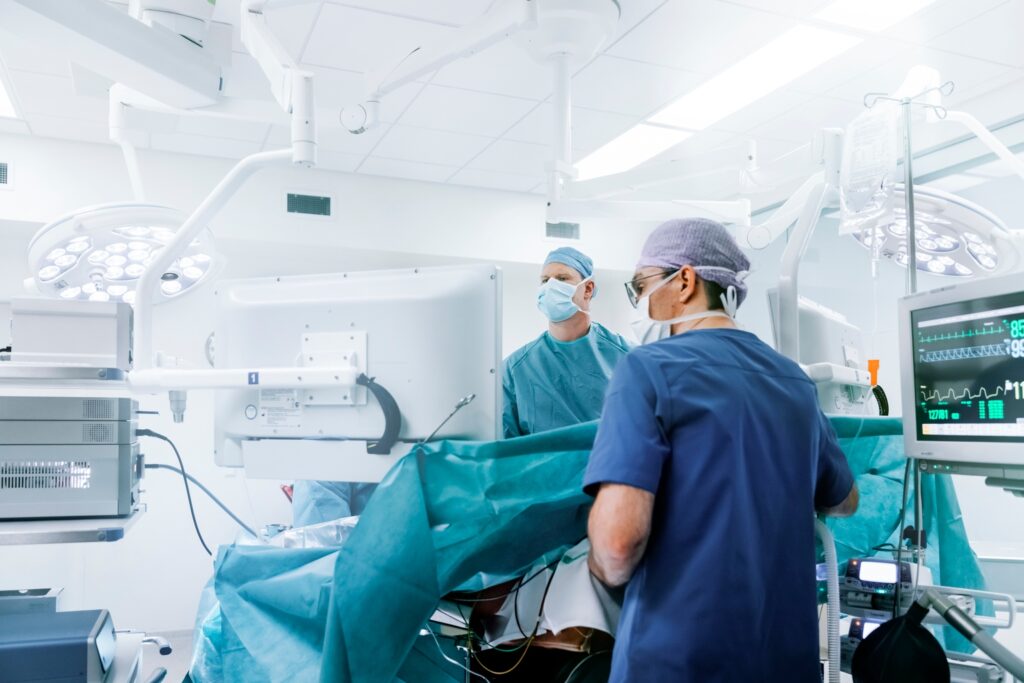
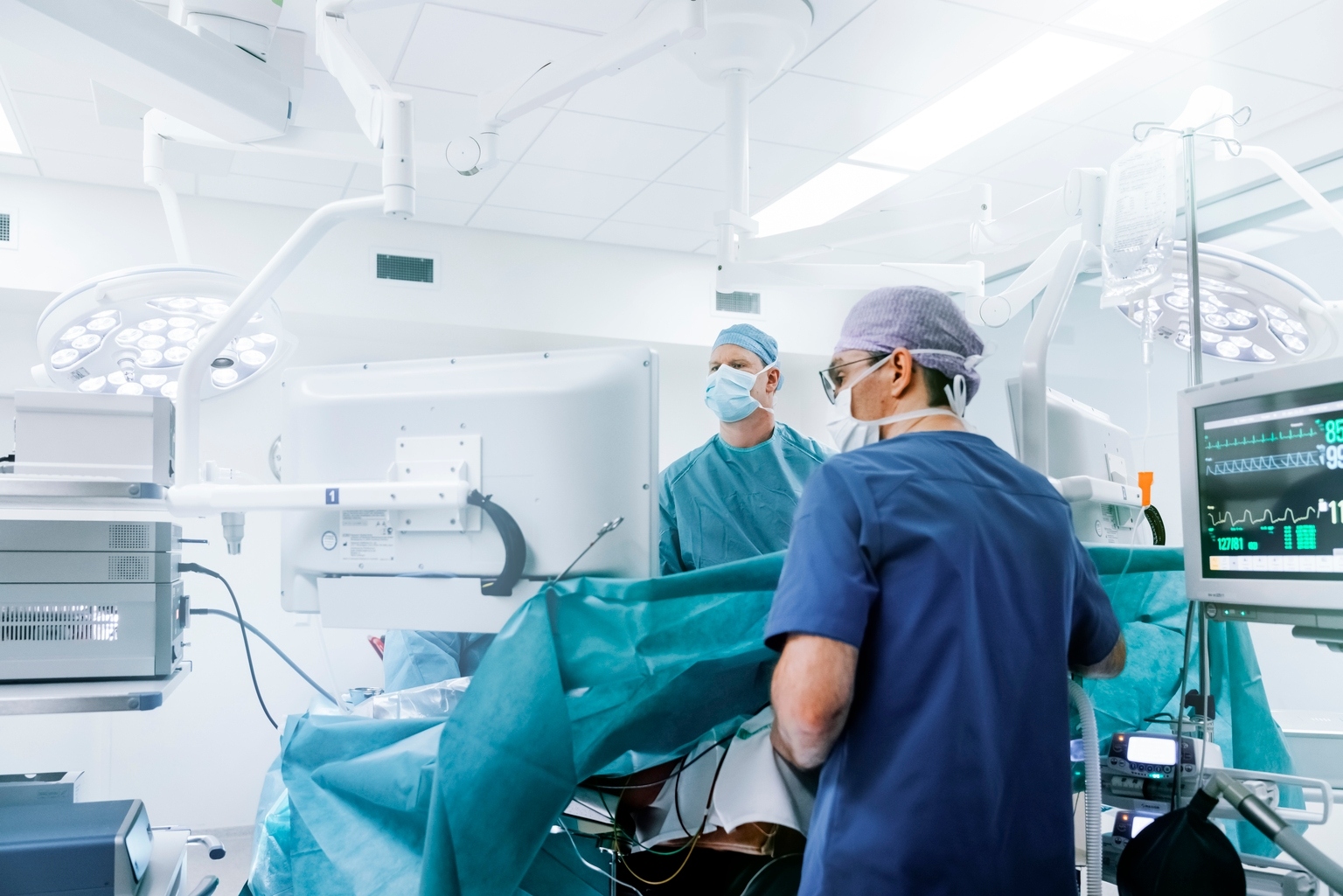
Integra LifeSciences’ CUSA Clarity gets FDA clearance for cardiac surgeries, enhancing debridement and valve repair.
Read the full article at: seekingalpha.com
Deep Learning Software for 3D Mammography Reconstruction Gets FDA Premarket Authorization | Diagnostic Imaging
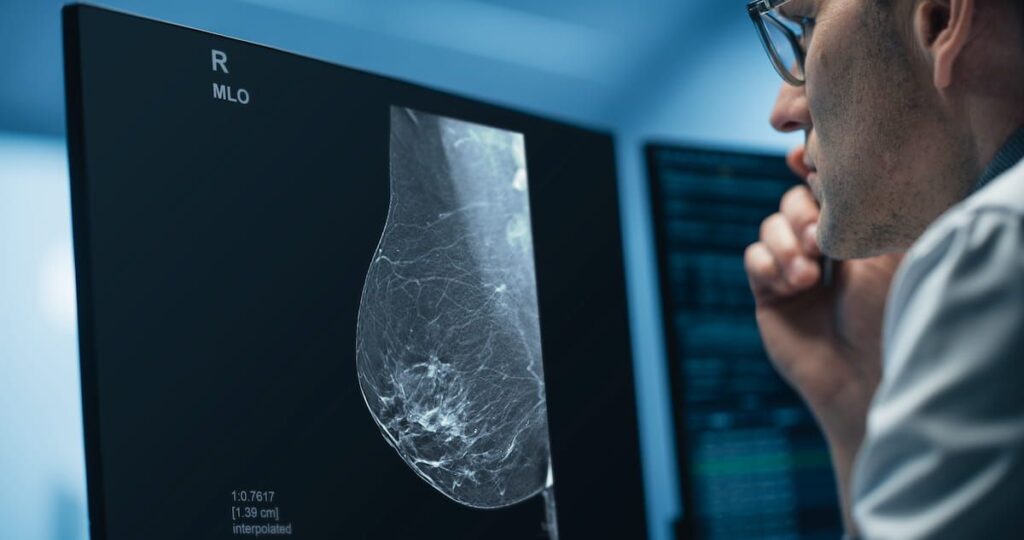
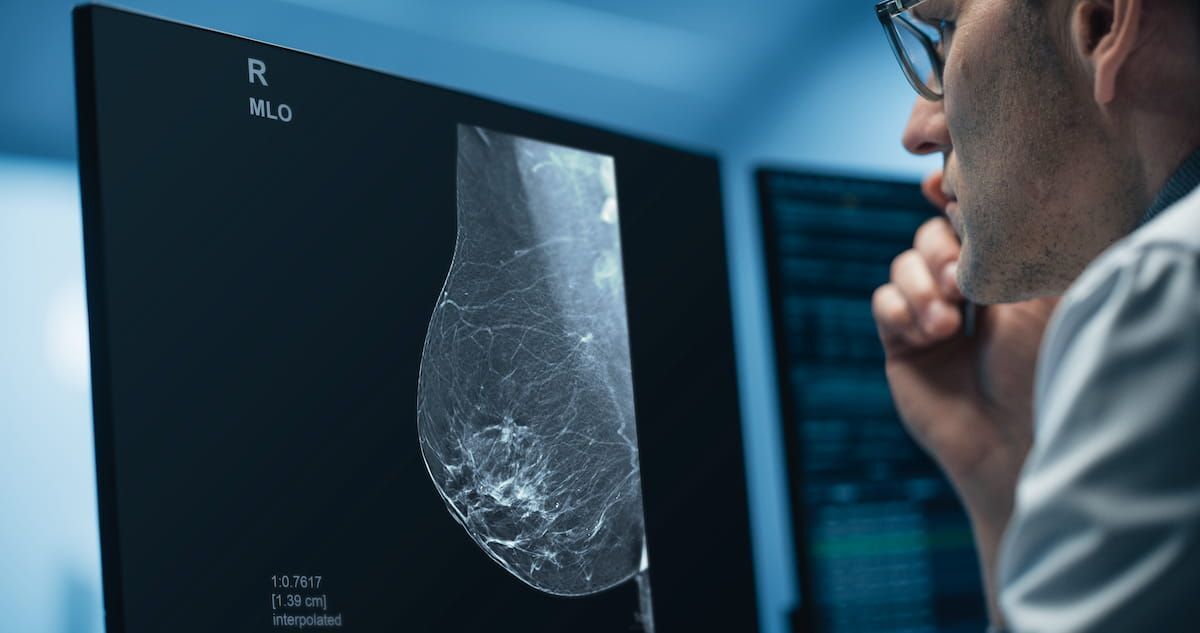
The combination of deep learning and iterative reconstruction with Pristina Recon DL may enhance image quality with digital breast tomosynthesis.
Read the full article at: www.diagnosticimaging.com
Cleveland Diagnostics wins FDA nod for prostate cancer test


The IsoPSA test is intended to help assess whether patients with elevated PSA levels should go on to receive a biopsy procedure.
Read the full article at: www.medtechdive.com
Fujifilm Sonosite Unveils Next-Generation Point-of-Care Ultrasound Device | Diagnostic Imaging
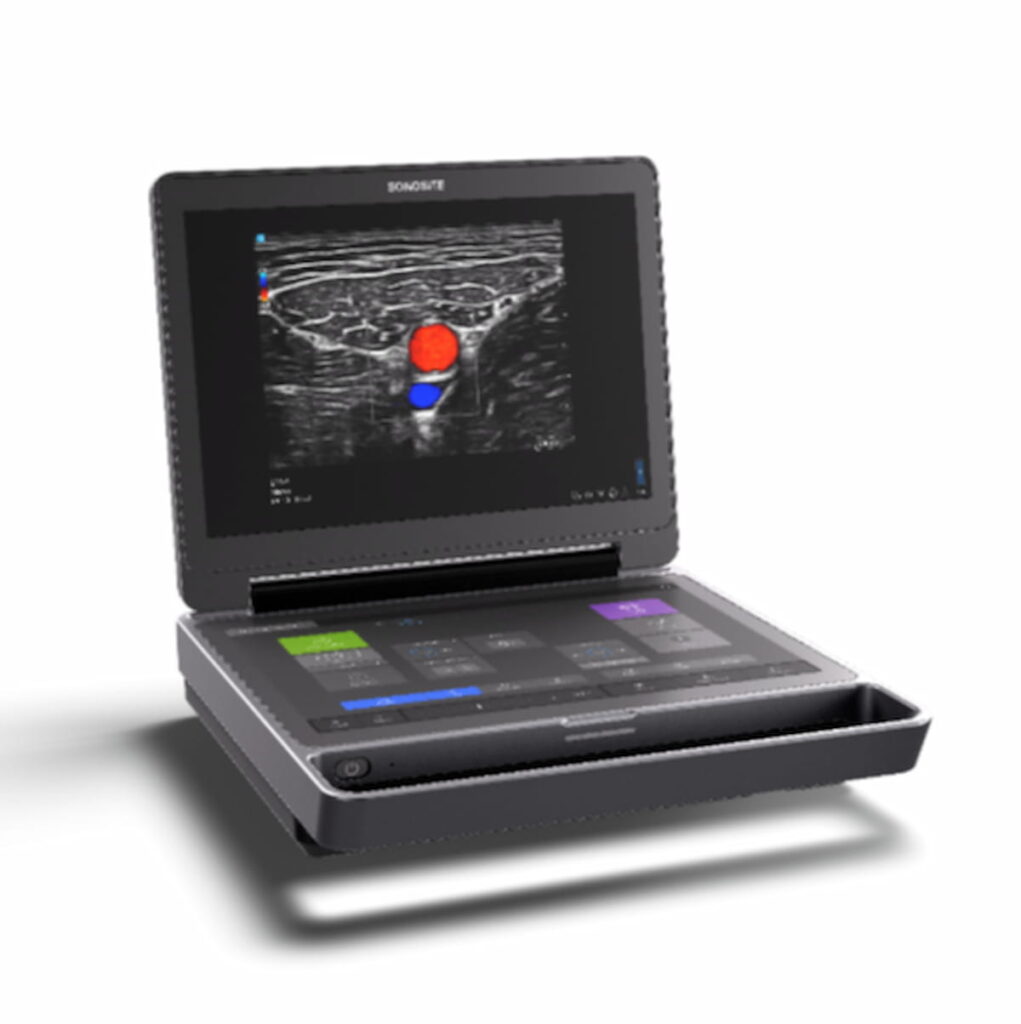
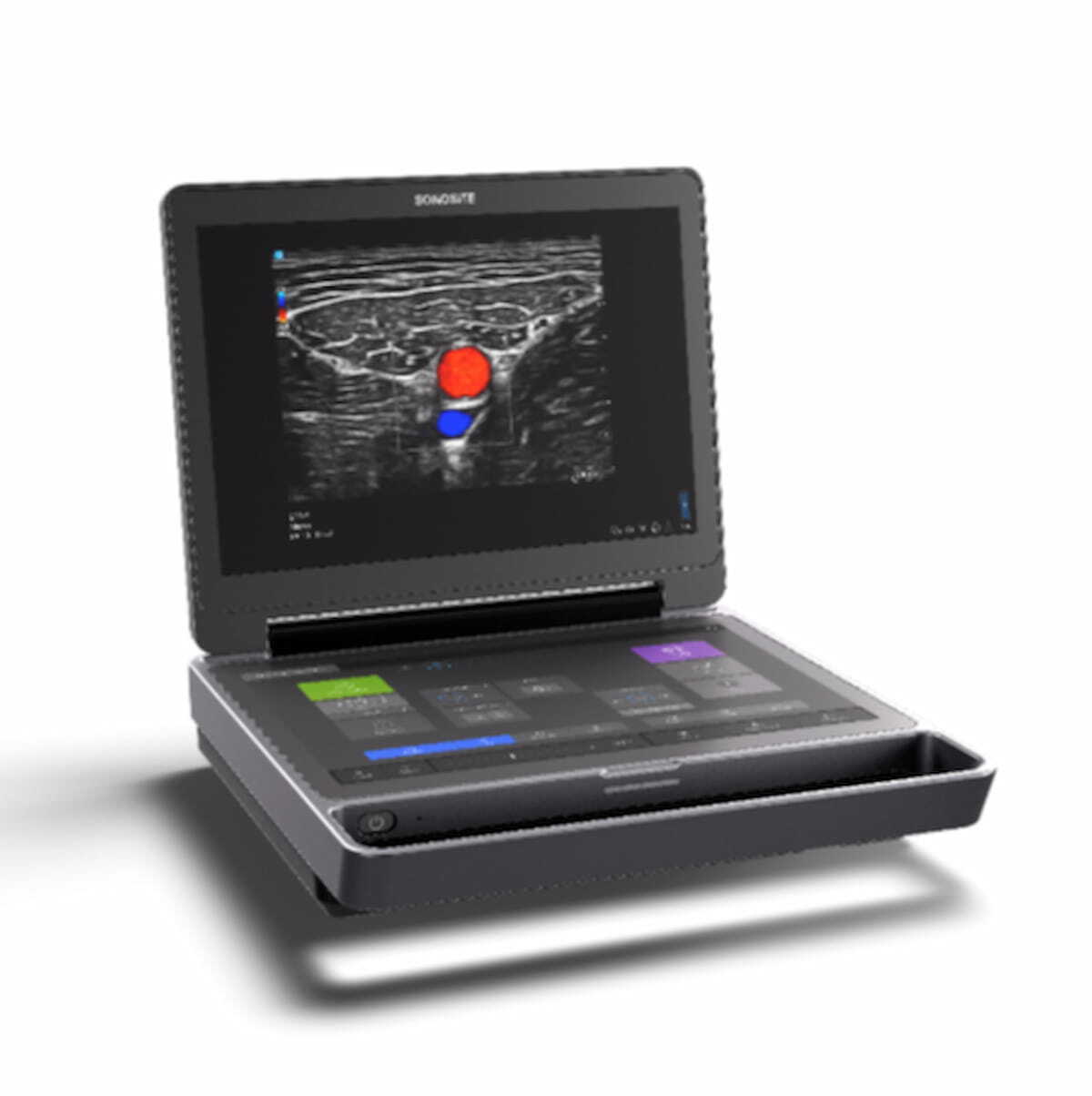
The Sonosite MT ultrasound device reportedly combines portability and enhanced high-resolution images with multiple features to promote workflow efficiency.
Read the full article at: www.diagnosticimaging.com
RapidAI Garners Four New FDA Clearances For CT-Based AI Modules | Diagnostic Imaging
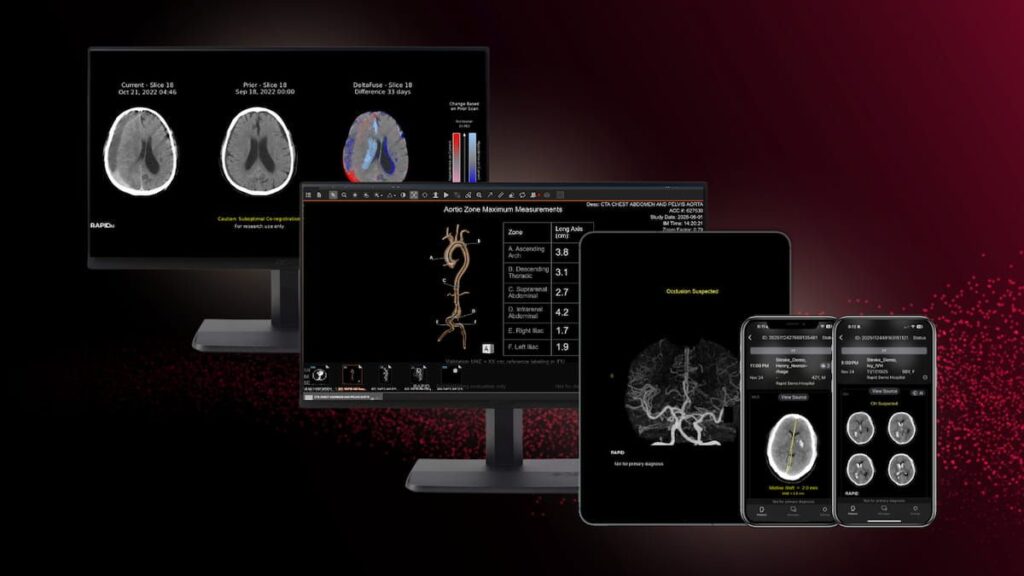
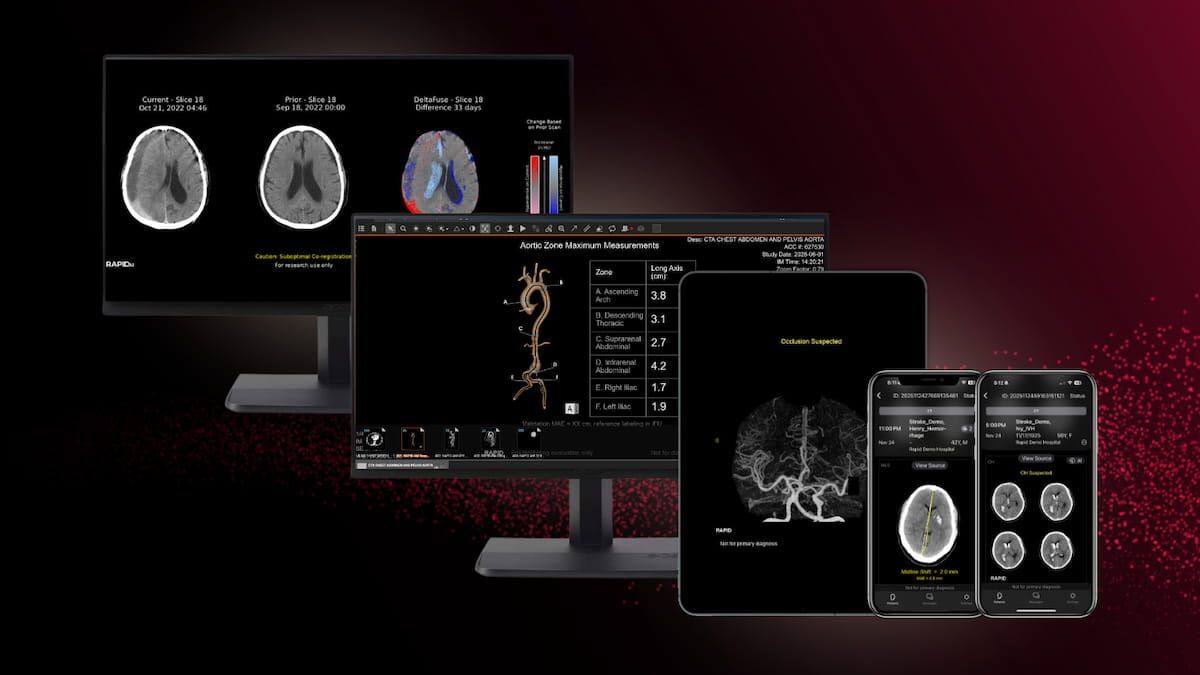
The new FDA 510(k) clearances for RapidAI include Rapid LMVO, which facilitates assessment of ischemic stroke, and Rapid MLS, which aids in the quantification of midline shifts with potential brain injuries.
Read the full article at: www.diagnosticimaging.com


