FDA Data Reveals Medical Device Recalls Software Woes


Discover how FDA medical device recall data exposes software design as a key factor in device recalls, impacting healthcare quality and patient assurance.
Read the full article at: spectrum.ieee.org
Ultrasound Renal Denervation System for Hypertension Garners CMS National Coverage Determination | Diagnostic Imaging
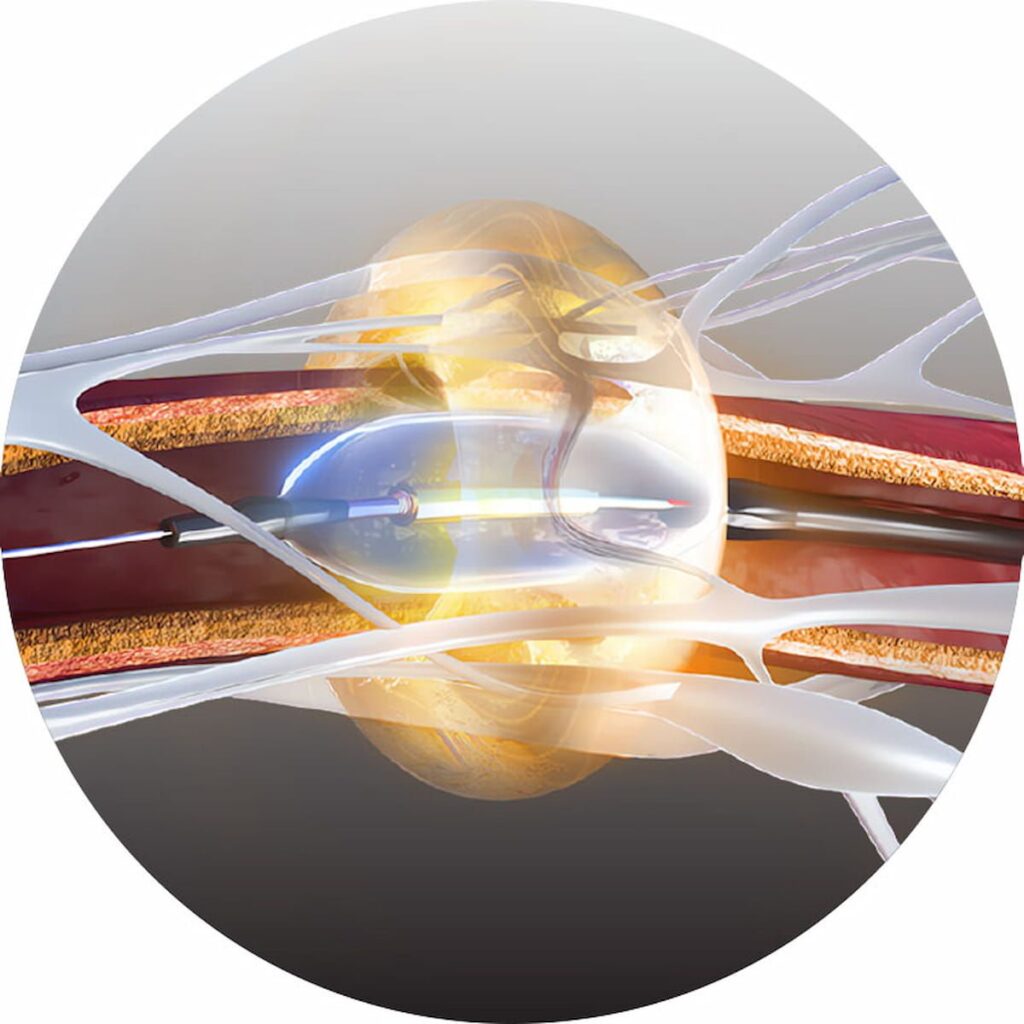
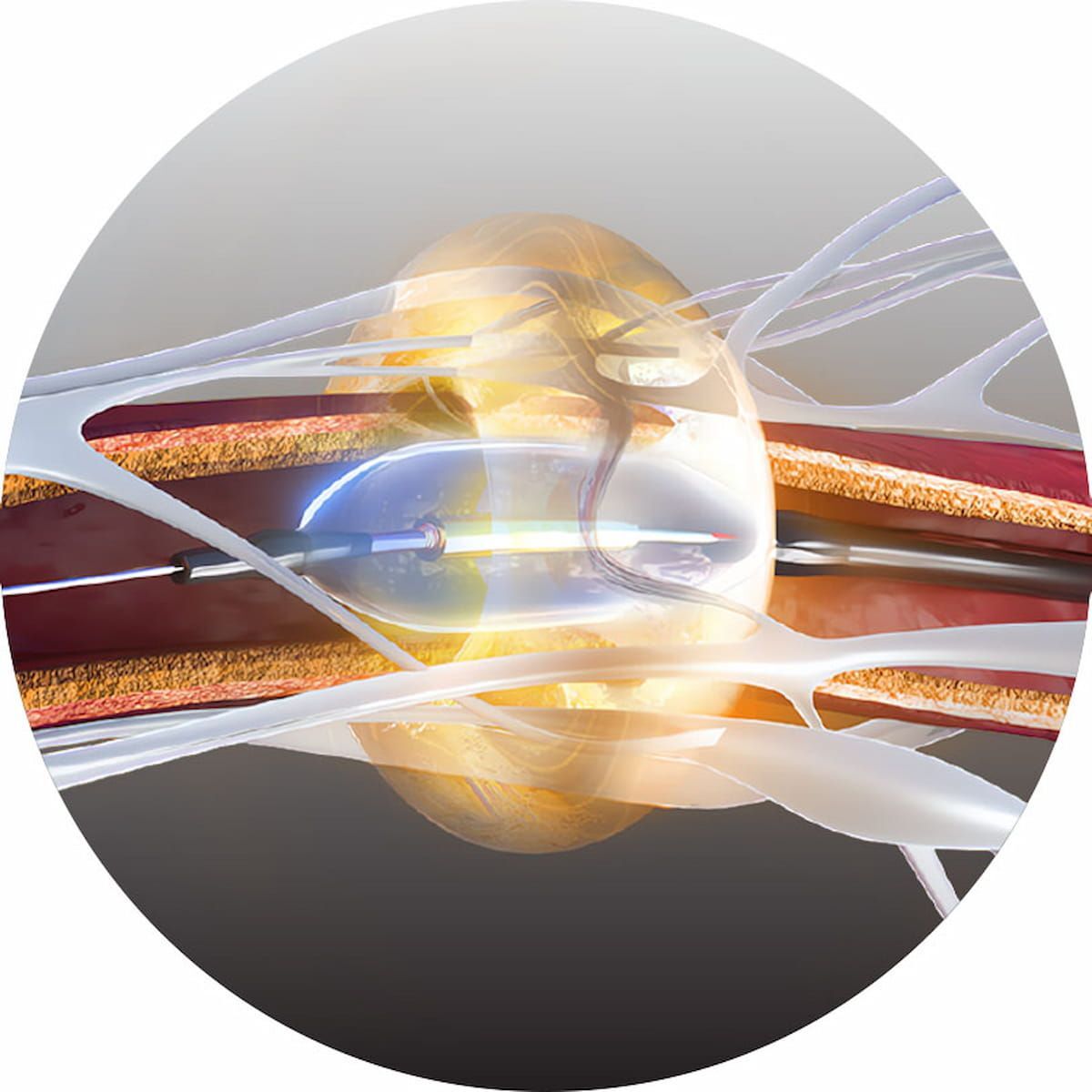
The Paradise Ultrasound Renal Denervation System has demonstrated efficacy and a favorable safety profile in randomized controlled trials for the treatment of mild, moderate and resistant hypertension.
Read the full article at: www.diagnosticimaging.com
Emerging AI Software for CT Plaque Analysis ‘Revolutionizes’ Carotid Artery Assessment | Diagnostic Imaging
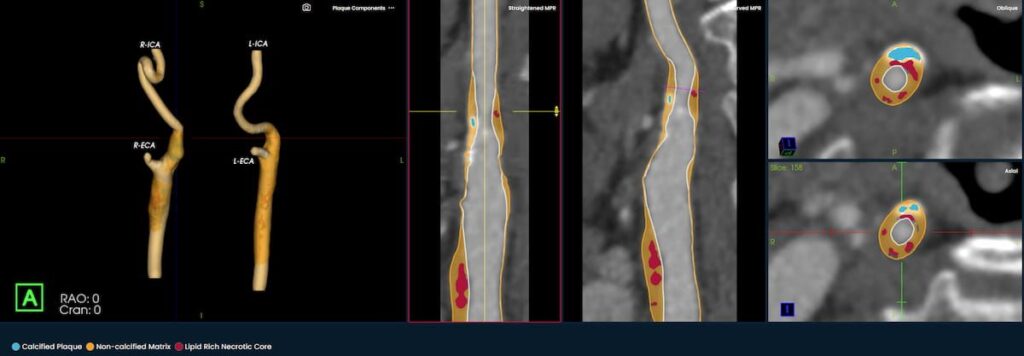
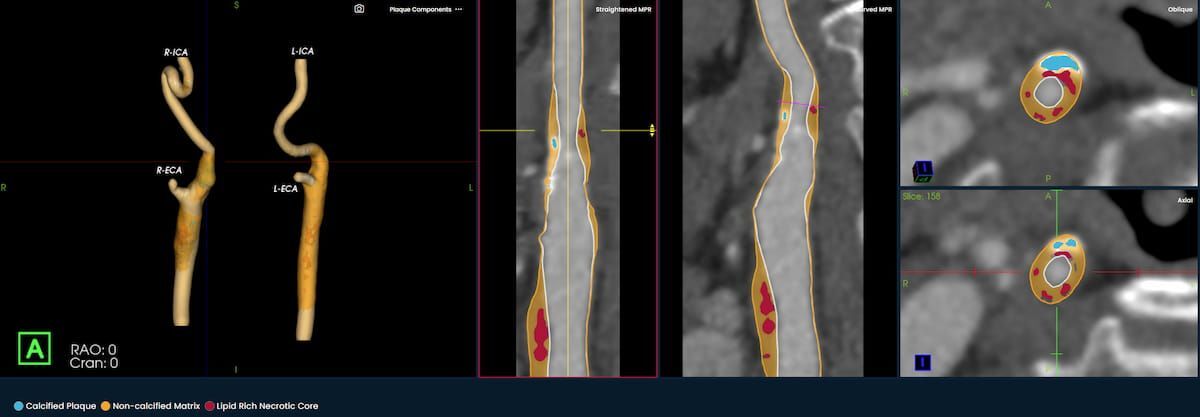
The newly launched PlaqueIQ™ image analysis modality is reportedly the first CT-based software indicated for the quantification and classification of plaque morphology in the carotid arteries.
Read the full article at: www.diagnosticimaging.com
BD Receives FDA 510(k) Clearance and CE-IVDR Certification for High-Throughput Enteric Bacterial Panels on BD COR™ System


/PRNewswire/ — BD (Becton, Dickinson and Company) (NYSE: BDX) a leading global medical technology company, today announced it has received U.S. Food and Drug…
Read the full article at: www.prnewswire.com
When Did the FDA Begin Regulating UVC Robots?
Infection Control Today serves infection control, facility, and C-suite leaders with strategies on HAIs, patient care, safety, and quality outcomes…
Read the full article at: www.infectioncontroltoday.com
QuidelOrtho Receives FDA 510(k) Clearance for VITROS™ Immunodiagnostic Products hs Troponin I Assay


/PRNewswire/ — The U.S. Food and Drug Administration (“FDA”) has granted QuidelOrtho Corporation (Nasdaq: QDEL) (“QuidelOrtho”), a global leader of in vitro…
Read the full article at: www.prnewswire.com
Tosoh Bioscience Receives FDA 510(k) Clearance for Fast, Accurate GR01 HbA1c Testing Analyzer


/PRNewswire/ — Tosoh Bioscience, Inc., a market leader in clinical diagnostics has received U.S. FDA 510(k) clearance for its next-generation Tosoh Automated…
Read the full article at: www.prnewswire.com
Best of the Best Skin Condition Diagnostic Tools 2026


Spread the loveIntroduction: The Evolution of Skin Diagnostics As we advance into 2026, the landscape of dermatological diagnostics has undergone a remarkable transformation. Driven by technological innovations and a deeper understanding of skin health, professionals now have access to a plethora…
Read the full article at: www.thetechedvocate.org
Terumo announces US FDA clearance for OpusWave imaging system

Terumo Interventional Systems has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its OpusWave dual-sensor imaging system.
Read the full article at: cardiovascularnews.com
Investor pressure may be driving risky AI medical device launches | Hub
Study finds an association between publicly traded companies and recalls of AI-based medical tools…
Read the full article at: hub.jhu.edu


