One-of-a-kind: Industrial design and engineering collaboration at Auburn University leads to groundbreaking medical device


/PRNewswire/ — Students from Auburn University’s Mechanical Engineering and Industrial Design programs recently witnessed something pretty special: the public…
Read the full article at: www.prnewswire.com
Dental measuring technology earns FDA clearance

Dental technology company Perimetrics has received FDA clearance for its InnerView System. The system is the first FDA-cleared technology that is designed to measure both internal and external mobility in teeth and implants, according to an Oct.
Read the full article at: www.beckersdental.com
FDA Grants De Novo Approval for Home Use of Prenatal Ultrasound Platform | Diagnostic Imaging
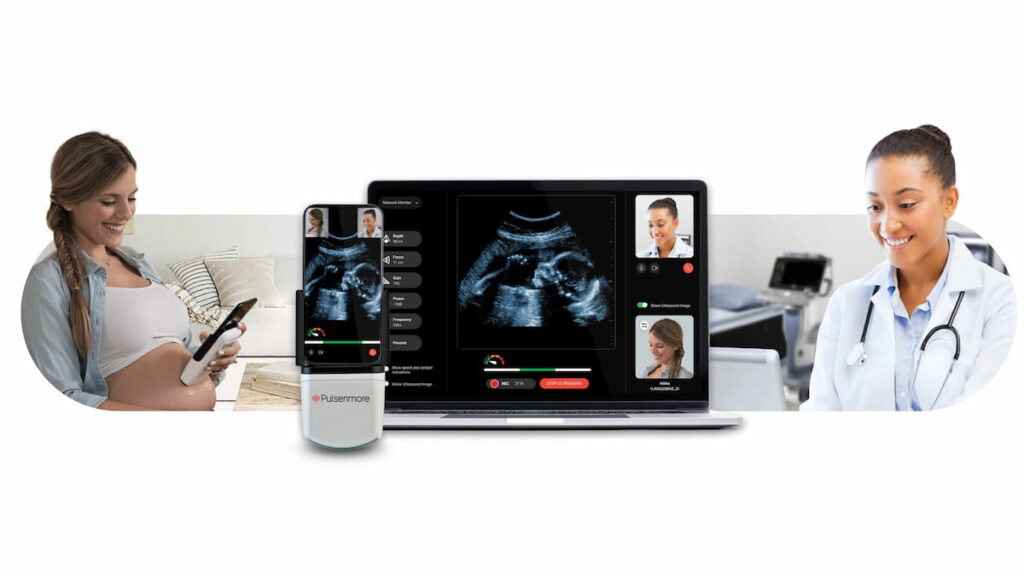

Already in use in multiple countries, the Pulsenmore ES ultrasound platform reportedly enables expectant mothers to transmit ultrasound video clips through a smartphone app for remote interpretation.
Read the full article at: www.diagnosticimaging.com
FDA Clears Plaque Assessment Software for Coronary CT Angiography | Diagnostic Imaging
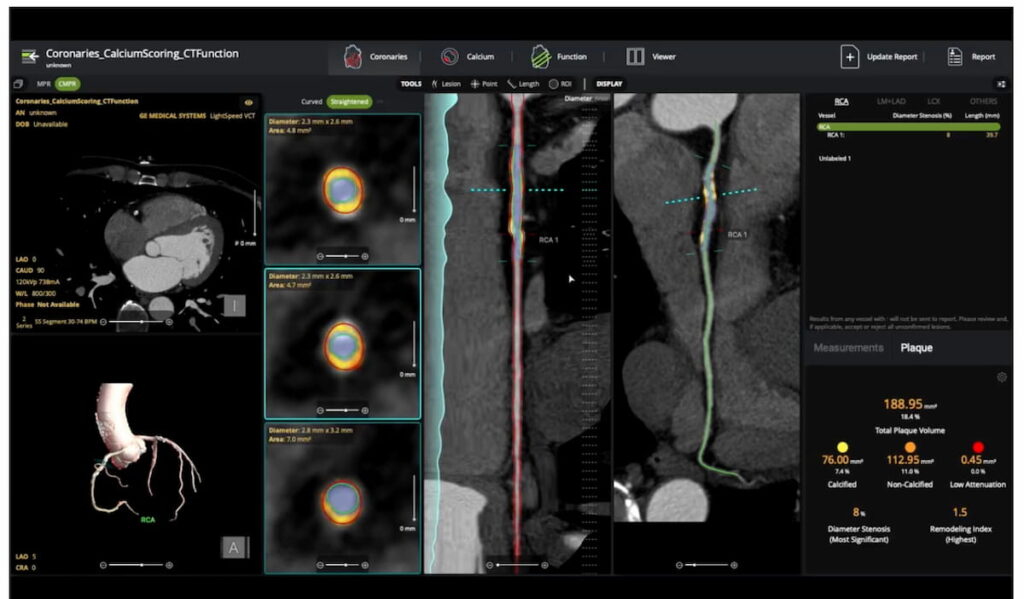

The AI-powered cvi42 | Plaque software enables adjunctive on-site interpretation of coronary CT angiography (CCTA) scans for plaque quantification.
Read the full article at: www.diagnosticimaging.com
Sirona Medical Gets FDA Nod for Advanced Imaging Suite | Diagnostic Imaging
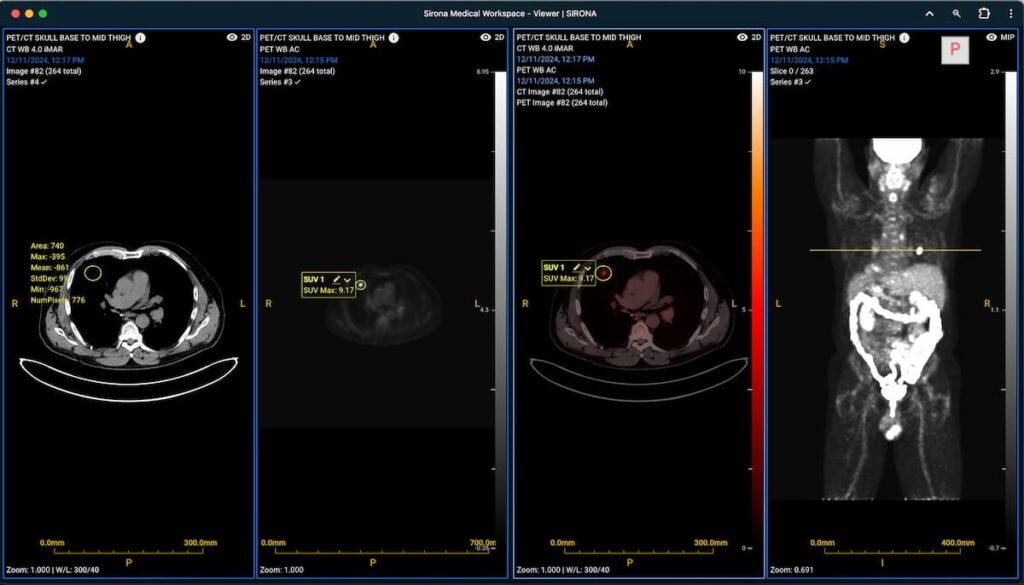

Expanded capabilities for the Sirona Advanced Imaging Suite reportedly include quantitative SUV analysis and multiplanar reconstruction for PET/CT imaging.
Read the full article at: www.diagnosticimaging.com
Mode Sensors Receives FDA 510(k) for Fluid Monitoring System
OSLO, Norway–(BUSINESS WIRE)–Mode Sensors (www.modesensors.com) announced today that its Re:Balans hydration monitoring system has received 510(k) clearance from the U.S. Food and Drug Administration, positioning the company to enter the American market with its wearable sensor technology. The clearance allows Re:Balans to be marketed in the United States for non-invasive monitoring of adult patients with fluid management-related health conditions, including those with fluid overload such as
Read the full article at: www.businesswire.com
FDA Grants HemoSonics 510(k) Clearance to Expand Use of Its Quantra Hemostasis System to Obstetric Procedures
DURHAM, N.C.–(BUSINESS WIRE)– #Childbirth–FDA Grants HemoSonics 510(k) Clearance to Expand Use of Its Quantra Hemostasis System to Obstetric Procedures
Read the full article at: www.businesswire.com
Hologic Receives FDA Clearance and CE Mark for Automated Molecular Tests to Detect Common Causes of Infectious Gastroenteritis
MARLBOROUGH, Mass.–(BUSINESS WIRE)—- $HOLX #Bacterial–Hologic, Inc. (Nasdaq: HOLX) announced today that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the Panther Fusion® Gastrointestinal (GI) Bacterial and Expanded Bacterial Assays. The company also announced that the assays obtained CE marking in the European Union in accordance with In Vitro Diagnostic Regulation (IVDR). These highly sensitive molecular tests are designed to rapidly detect the most common bacterial pathogens res
Read the full article at: www.businesswire.com
Synchrony Medical Launches LibAirty™ Airway Clearance System in the U.S., Offering a New Standard for At-Home Respiratory Care


/PRNewswire/ — Synchrony Medical, a medical technology company dedicated to transforming respiratory care, announced today the launch of its LibAirty™ Airway…
Read the full article at: www.prnewswire.com
Biolinq gets FDA de novo nod for intradermal glucose sensor


The device, called Biolinq Shine, is indicated for people with Type 2 diabetes who don’t depend on insulin.
Read the full article at: www.medtechdive.com


