Smartlens, Inc. Secures $5.2 Million Bridge Round to Advance FDA Clearance Process and Prepare for Commercial Launch of miLens


/PRNewswire/ — Smartlens, Inc., a clinical-stage ophthalmic technology company advancing next-generation glaucoma management devices, today announced the…
Read the full article at: www.prnewswire.com
Auxano® Medical Receives FDA 510(k) Clearance for the ARKEO™ Wedge Fixation System – Ortho Spine News


An important milestone in the company’s mission to deliver precision-engineered orthopedic solutions BRECKSVILLE, OH, UNITED STATES, October 16, 2025 /EINPresswire.com/ — Auxano® Medical is proud to announce its wedge fixation system, ARKEO™, has received U.S. Food and Drug Administration (FDA) 510(k) clearance, marking an important milestone in the company’s mission to deliver precision-engineered orthopedic solutions that empower surgeons and improve patient outcomes. The ARKEO™ Wedge Fixation System integrates an anatomical design with the pillared microstructure of the Auxano® in-growth technology to provide a secure fixation interface at the site of correction. Designed for use in Evans and Cotton osteotomies, the ARKEO™ surgical set includes intuitive instrumentation that enhances reproducibility by giving surgeons complete control over implant positioning throughout the procedure. “Achieving 510(k) clearance for ARKEO™ represents a significant advancement in our orthopedic portfolio,” said Joe Harr, Sales & Distribution Manager at Auxano® Medical. “This system combines modern engineering with practical, surgeon-focused innovation to deliver reproducible results and improve the surgical experience.” The ARKEO™ system is designed to optimize fixation and graft integration through several key features: • Anatomical Fit: Subflush placement reduces irritation and maintains alignment. • Open Architecture: Large central graft cavity and thru-growth fenestrations maximize graft capacity and promote biologic fixation. • Controlled Stability: The unique pillared surface morphology has demonstrated viability in scientific research studies and clinical efficacy in midfoot fusion applications. • Intuitive Instrumentation: Simplifies each step for efficient, consistent outcomes. Supplied non-sterile in a steam-sterilizable tray, the ARKEO™ system includes a Hintermann distractor, k-wires, osteotomes, mallet, slap hammer, implant trials, inserter, and an impactor. “The ARKEO™ Wedge Fixation System exemplifies Auxano® Medical’s commitment to designing devices that are both clinically advanced and intuitively engineered,” shared Dr. George Picha, Ph.D, F.A.C.S. “With this clearance, we are proud to continue expanding our solutions for orthopedic surgeons across the U.S.” For more information about the ARKEO™ Wedge Fixation System, visit www.AuxanoMedical.net or contact CS@AuxanoMedical.net.About Auxano® Medical Auxano® Medical develops and manufactures advanced orthopedic solutions that support improved patient outcomes and procedural efficiency. With a commitment to quality, innovation, and clinical collaboration, Auxano® delivers technologies that meet the highest standards of performance and reliability. The company is headquartered near Cleveland, OH. For more information, visit www.auxanomedical.net Halle SchosslerAuxano Medicalemail us here Legal Disclaimer: EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Read the full article at: orthospinenews.com
FDA Green Lights Ruthlessly Practical ‘Smart’ Device

Spine surgeons know the drill: precision is everything when it comes to pedicle screw placement.
Read the full article at: ryortho.com
FDA halts new device submissions during government shutdown


The FDA will not accept new medical device submissions that require payment of user fees during the Trump administration’s government shutdown.
Read the full article at: www.medicaldesignandoutsourcing.com
Next-Generation AI-Driven SyncAR® Spine Receives FDA Clearance for Spine Surgery –
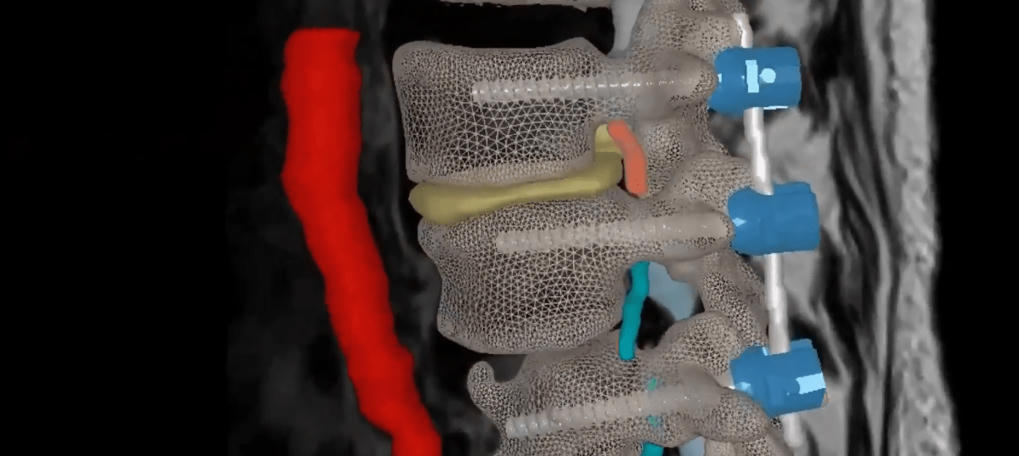

Surgical Theater, the leader in surgical XR visualization, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for SyncAR® Spine, the next-generation release of its spine platform.
Read the full article at: orthofeed.com
Versatile Full-Body Cone-Beam CT Scanner Gets FDA Clearance

The Planmed XFI full-body weightbearing cone-beam CT scanner reportedly enables upright and supine patient positioning.
Read the full article at: www.diagnosticimaging.com
Prytime Medical Devices Receives World’s First Extended Duration FDA 510(k) Clearance for pREBOA-PRO™ Catheter


/PRNewswire/ — Building once again on their first mover position as a leader in endovascular hemorrhage control and resuscitation, Prytime Medical Devices,…
Read the full article at: www.prnewswire.com
Top 3D Printing Applications in Medical Device Production


3D Printing has become a transformative force in medical device production, offering unprecedented flexibility, customization, and precision. As the technology rapidly advances in 2025, medical device manufacturers are adopting it not just for prototype development but also for producing fully functional medical devices tailored to individual patients. This blog explores the top 3D printing applications reshaping medical device manufacturing today, highlighting innovations that improve surgical outcomes, reduce costs, and unlock new possibilities in personalized patient care. What we cover in this blog? Top Applications of 3D Printing in Medical Device Production Top Applications of 3D Printing in Medical Device Production 1. Custom Implants and Prosthetics One of the significant uses of 3D printing in medical device production is the creation of custom implants and prosthetics. Unlike traditional manufacturing processes that rely on standardized designs, 3D printing enables the fabrication of implants designed precisely to the patient’s unique anatomy using advanced imaging data such as CT or MRI scans. Breakthroughs in Implant Technologies In 2025, advancements in additive manufacturing will allow for the use of bioresorbable materials and shape-memory alloys in implants. For example, researchers have developed 3D-printed heart valves made from shape-memory materials that can adapt post-implantation while gradually being absorbed by the body, thereby reducing the need for repeated surgeries in pediatric patients. Orthopedic devices, such as patient-specific talus spacers for ankle reconstruction, are also being widely produced through 3D printing, offering joint-sparing options that improve pain relief and mobility. Benefits for Patients and Surgeons Custom implants improve surgical accuracy by perfectly matching complex bone geometries, which leads to fewer complications, shorter recovery times, and better integration with existing tissues. Surgeons gain the ability to plan and rehearse procedures using patient-specific 3D models, enhancing precision and confidence during complex surgeries. 2. Surgical Guides and Planning Models 3D printing plays a crucial role in preoperative surgical planning by producing accurate anatomical models and cutting guides. These patient-specific models allow surgeons to visualize complex anatomies, practice interventions, and develop precise surgical strategies tailored to the individual’s needs. Expansion Across Specialties While orthopedic and cranio-maxillofacial surgeries were early adopters, 2025 is expected to see a significant increase in adoption across specialties such as congenital heart disease and oncology. Patient-specific models used in congenital heart defect surgeries help reduce ICU time and improve outcomes by giving surgeons a tactile, highly detailed reference. Oncology teams benefit from 3D printed guides for tumor resections, facilitating precise radiotherapy targeting and sparing healthy tissue. The Best Contract Manufacturers in India are renowned for delivering high-quality products, innovative solutions, and reliable services across industries. Operational Efficiency Gains Hospitals are increasingly integrating 3D printing directly into their point-of-care facilities, reducing dependence on external providers. Trends toward automation, AI-supported segmentation, and extended reality tools enhance workflow efficiency, allowing more patients to access these innovative aids more quickly and effectively. The ability to quickly generate these models reduces procedure times and postoperative complications. 3. Biocompatible Medical Devices and Wearables The third major application of 3D printing in medical device manufacturing is the production of biocompatible devices and wearable medical technology. This encompasses everything from dental applications and orthoses to wearable sensors and bioactive implants. Dental and Orthopedic Devices The dental sector is booming, with 3D printed crowns, bridges, aligners, and mandibular reconstruction plates becoming standard due to faster, more precise manufacturing processes. Materials like PEEK and biocompatible polymers enable durable, lightweight medical wearables and braces tailored perfectly to patient morphology. Smart Implants and Embedded Sensors Innovations in materials science have introduced hybrid polymers and nanostructured surfaces that promote faster tissue integration, reduce infection risks, and enable the embedding of electronics. 3D printing allows the creation of implants with integrated sensors that can monitor parameters such as pressure, strain, or biochemical markers in real-time, paving the way for next-generation personalized therapies and remote health monitoring. Market Growth and the Road Ahead The medical 3D printing industry is witnessing explosive growth. Market projections estimate the global 3D printing medical devices market will surge from $4.37 billion in 2025 to nearly $19 billion by 2035, fueled by increasing adoption of personalized devices and new regulatory clarity encouraging innovative biocompatible materials. Several factors are driving this momentum: Growing demand for patient-specific implants and devices Expansion of in-hospital and point-of-care 3D printing capabilities Integration of AI for design and operational efficiencies Advances in biocompatible printing materials and multi-material printing Challenges remain, including ensuring quality control, sterilization validation, and scalable manufacturing; however, ongoing research and regulatory focus aim to overcome these issues. A ISO 13485:2016 certified company for manufacturing medical equipment and components, offers expertise in 3D printing. Conclusion 3D printing is both a prototyping tool and a core enabler of next-generation medical device manufacturing. Its applications in custom implants, surgical planning models, and biocompatible wearables are revolutionizing patient care by delivering highly personalized, precise, and efficient solutions. Medical device manufacturers and healthcare providers embracing these innovations today are setting the stage for a future where tailored treatments and rapid innovation improve outcomes and patient experiences worldwide. As 3D printing technology advances, the possibilities for medical applications will continue to expand, making personalized medicine more accessible and transforming healthcare as we know it. FAQs 1. What are the most common uses of 3D printing in medical devices? 3D printing is widely used to create custom implants, surgical guides, prosthetics, and rapid prototypes in medical device manufacturing. 2. Can entire medical devices be 3D printed? Yes, many 3D printed devices like dental restorations, hearing aids, and orthopedic supports are fully functional and patient specific. 3. What materials are used for 3D printing medical devices? Medical-grade biocompatible materials include resins, titanium alloys, and specialized polymers suitable for implants and surgical tools. 4. Is 3D printing safe and regulated for medical applications? When done with certified materials and processes, 3D printing is safe; regulatory bodies like the FDA provide guidance and clearances for many printed devices. The post Top 3D Printing Applications in Medical Device Production appeared first on Violin Technologies.
Read the full article at: www.blogarama.com
Nurea Receives FDA Clearance for PRAEVAorta 2


AI-powered software helps vascular physicians by supporting diagnostic decision-making and patient follow-up, improving reliability and accuracy while reducing the cost of aneurysm care.
Read the full article at: www.dicardiology.com


